Market Trends of Zika Virus Testing Industry
Molecular Test Segment is Expected to Show Better Growth Over the Forecast Years
Nucleic Acid Amplification Test (NAAT) is a generic term referring to all molecular tests used to detect viral genomic material. The NAAT assays are the preferred method of diagnosis because they can provide confirmed evidence of infection. The increasing demand for tests for zika virus testing has been increasing recently. Hence the molecular test segment is anticipated to witness growth over the forecast period.
The increasing research and development in the molecular testing segment are bolstering the growth of the segment. For instance, in March 2022, an international team of researchers headed by experts from the University of Toronto revealed results for one of the first field trials for a synthetic biology-based diagnostic using patient samples. This study was conducted on-site in Latin America and reveals the potential for cell-free synthetic biology tools and companion hardware for providing rapid, decentralized, and low-cost patient testing for infectious diseases like the Zika virus.
The study results published in Nature Biomedical Engineering in March 2022 showed that the novel diagnostic platform has analytical specificity and sensitivity, which is equivalent to the United States Centre for Disease Control PCR test for Zika, and has a diagnostic accuracy of 98.5% with 268 patients samples collected in Recife, Brazil. The platform is also programmable and can be similarly applied to detect any pathogen sequence. Also, the researchers state that on the molecular side, the cell-free tests can be freeze-dried, allowing for distribution without refrigeration. All the molecular components of the test are independent of the PCR-supply chain. Thus, the rising research on molecular testing and its efficacy is bolstering segment growth.
The increasing adoption of molecular Zika virus testing by various government organizations and market players' strategies, such as partnerships and acquisitions, are also expected to contribute to the growth of the studied segment. For instance, in November 2021, the India Molecular Diagnostics and Research Laboratory (MDRL) of the Government Institute of Medical Sciences (GIMS) in Greater Noida, India, started conducting tests for the zika virus. Also, in September 2021, Roche acquired 100% of the shares of TIB Molbiol Group to enhance its molecular diagnostic solutions portfolio.
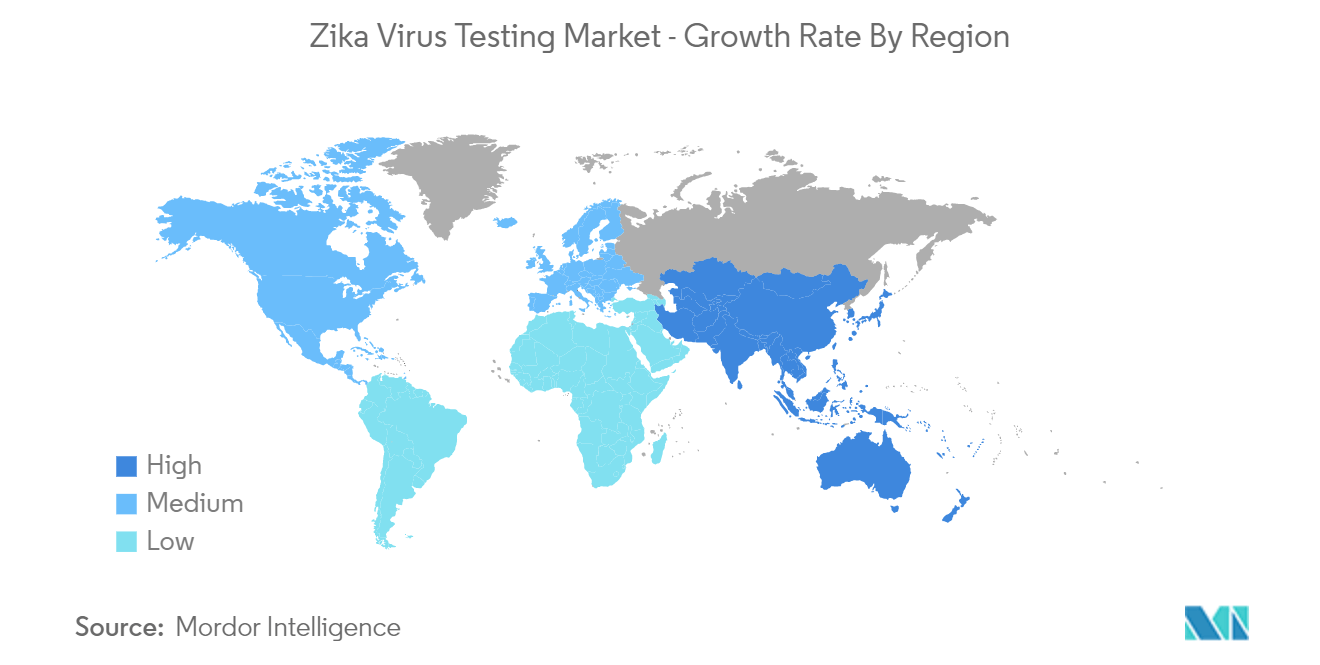
Asia Pacific is Expected to Hold Significant Share in the Market Over the Forecast Period
Asia Pacific is expected to hold a significant share of the market over the forecast period. Factors such as the increasing prevalence of the Zika virus (ZIKV) in countries across Asia-pacific and rising demand for effective testing are expected to contribute to the growth of the market in this region over the forecast period.
The prevalence and incidence of the zika virus in India are anticipated to drive the demand for zika virus testing in the country. For instance, the WHO data updated in October 2021 showed that on July 8, 2021, a ZIKV infection was laboratory-confirmed in a resident of Kerala, India. This represented the first Zika virus disease case ever reported from Kerala. Again, on July 31, 2021, Maharashtra state also reported its first Zika laboratory-confirmed case from Belsar village in Pune, India. In addition, Indian health authorities reported a total of 152 Zika virus cases in Uttar Pradesh as of December 5, 2021. The outbreak began in October and represented the first major outbreak identified in the state. Such incidence of zika virus cases in Asia-Pacific countries is expected to contribute to the demand for Zika virus testing, thereby boosting the growth of the market.
Moreover, as per the data provided by the Union Minister of Health and Family Welfare in December 2021, 237 Zika virus cases were reported in December 2021 across India. Hence, the increasing prevalence of the zika virus has created an emergence of zika virus testing in this region, thereby driving the growth of the studied market in this region.
Likewise, serological evidence of widespread Zika Virus has been found in some other countries in Asia-Pacific. An NCBI article published in July 2021 stated that when 997 serum samples were randomly collected from dengue patients and tested for ZIKV, widespread ZIKV exposure was found in the Philippines. The above-mentioned study also suggested that zika virus testing across the country is needed to find the ZIKV infection risk factor. Thus, the spread of the zika virus across countries in Asia-pacific is expected to drive the studied market growth in the region.
Therefore, owing to the factors mentioned above, such as the high prevalence of zika virus infections and the rising demand for effective diagnosis, the zika virus testing market is anticipated to see growth over the forecast period in the Asia-Pacific region.