Market Trends of Vascular Closure Device (VCD) Industry
This section covers the major market trends shaping the Vascular Closure Device (VCD) Market according to our research experts:
Femoral Access Segment is Expected to Show Significant Growth Over the Forecast Period
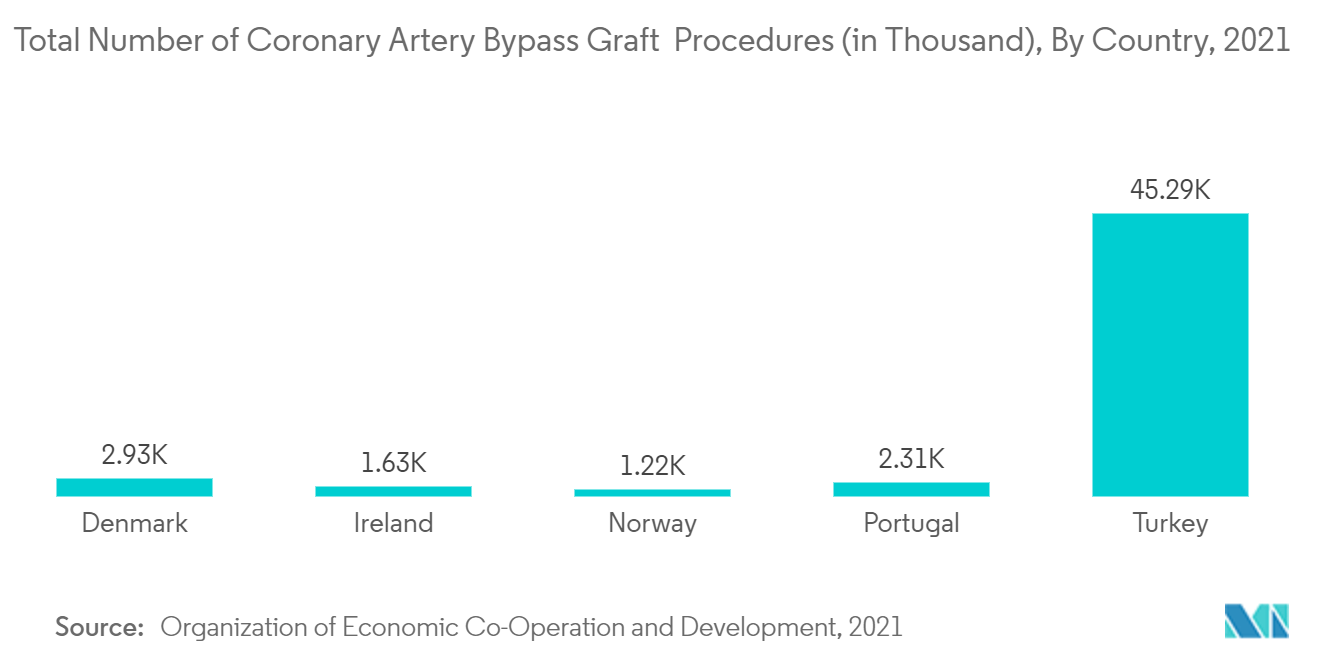
Femoral vascular closure devices are powerful tools for interventional cardiologists and other proceduralists to achieve hemostasis at the catheterization entry site. The market segment growth is boosted by the research studies demonstrating the benefits of femoral vascular closure devices. For instance, an article titled 'Comparison of MynxGrip vascular closure device and manual compression for closure after femoral access angiography: a randomized controlled trial: the closure devices used in everyday practice study, CLOSE-UP III trial' published in the journal of BMC Cardiovascular Disorders in February 2022 compared the safety and efficacy of two vascular closure devices available in the market. The study concluded that the occurrence of a major adverse vascular event was rare in both cases. Such benefits of the femoral access vascular closure devices promote their usage, hence driving the growth of the market segment.
Additionally, the collaboration among the market players for the marketing and commercialization of devices in different regions is driving the growth of the market segment. For Instance, in May 2022, Teleflex Incorporated, a medical technologies provider, received Health Canada approval for the MANTA Vascular Closure Device, the first commercially available biomechanical vascular closure device designed specifically for large bore femoral arterial access site closure. MANTA Device is indicated for closure of femoral arterial access sites while reducing time to hemostasis following the use of 10-20F devices or sheaths (12-25F OD) in endovascular catheterization procedures.
Thus, owing to the abovementioned factors, the market segment is expected to grow significantly over the forecast period.
North America Holds Major Share in the Market and is Expected to do the Same Over the Forecast Period.
The well-established healthcare infrastructure, the growing burden of cardiovascular diseases, coupled with an increase in catheterization-related procedures, and the rise in demand for minimally invasive solutions in the country are driving the growth of the market in the region. According to the Center for Disease Control and Prevention (CDC) article titled 'Heart Diseases Facts' updated in February 2022, heart disease is the leading cause of death in the United States. It also reported that about 659,000 people in the United States die from heart disease each year. Such a high burden of the disease generates the need for vascular closure devices and drives the growth of the market in the region.
Additionally, the market growth is also boosted by approval from regulatory authorities such as the United States Food and Drug Administration and Health Canada. For instance, in April 2021, EnsiteVascular received its second United States Food and Drug Administration (FDA) market clearance for the company's SiteSeal SV (small vessel) VCD compression device to achieve hemostasis of the brachial artery after a transbronchial endovascular procedure. SiteSeal SV is a new closure device designed for the closure of small arterial vessels, specifically brachial, radial and pedal. It simulates external compression, but removes the associated variables, leaving nothing behind.
Also, in October 2021, Haemonetics received approval from the United States Food and Drug Administration for same-day discharge after atrial fibrillation ablation using its Vascade MVP venous vascular closure system. Vascade MVP is a vascular closure system that is designed for procedures that require multiple access site venous closures.
Thus, owing to the abovementioned factors, the vascular closure device market is expected to project significant growth in the North American region over the forecast period.