Vascular Access Devices Market Analysis by Mordor Intelligence
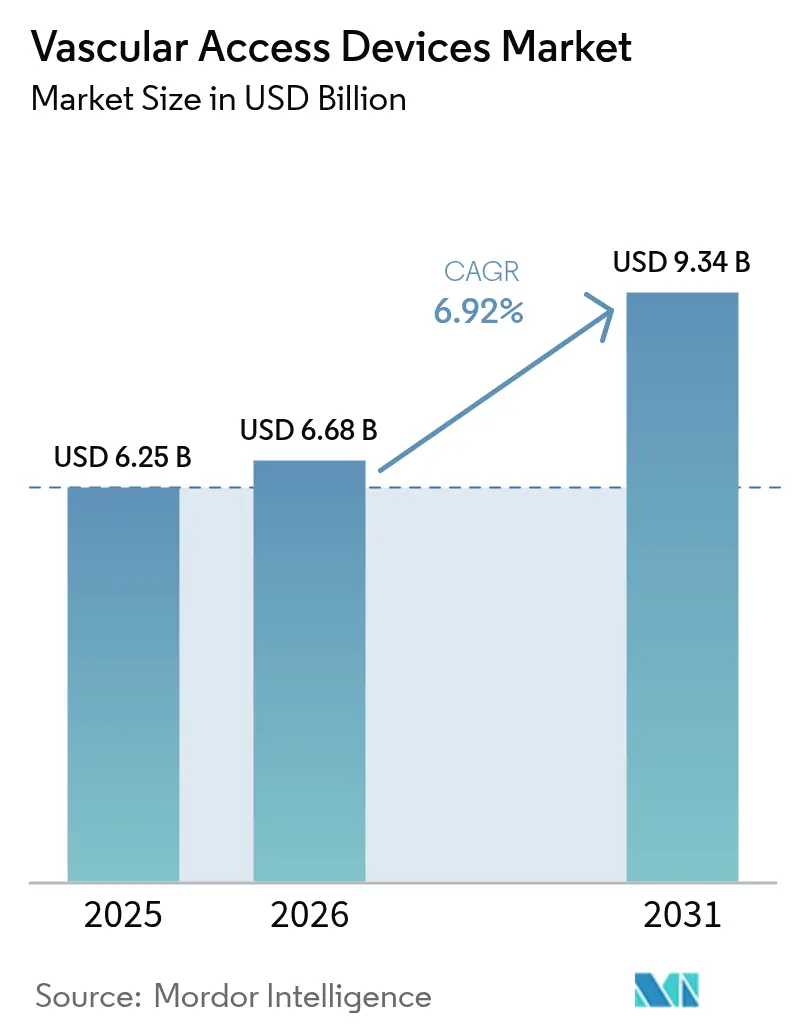
The vascular access devices market size is expected to grow from USD 6.25 billion in 2025 to USD 6.68 billion in 2026 and is forecast to reach USD 9.34 billion by 2031 at 6.92% CAGR over 2026-2031. This expansion reflects healthcare providers’ pivot toward value-based innovation that prioritizes infection prevention, material durability and procedural efficiency. A rising chronic disease burden, broader adoption of ultrasound-guided insertion and the shift to outpatient care models collectively underpin sustained demand. At the same time, advances in hydrophilic biomaterials and antimicrobial coatings intensify product differentiation, while supply-chain reshoring helps manufacturers mitigate raw-material risk. Competitive dynamics therefore favor firms able to pair scale manufacturing with rapid technology roll-outs, keeping the vascular access devices market on a steady growth trajectory.
Key Report Takeaways
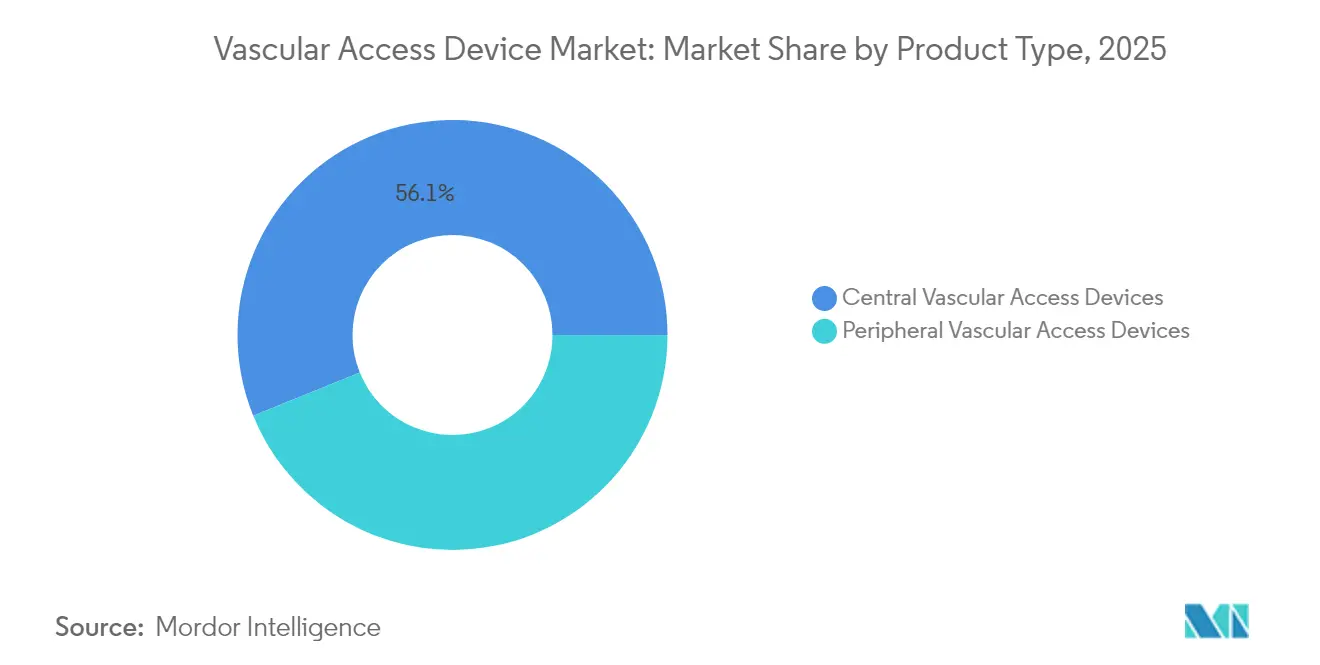
- By device type, central vascular access devices led with 56.12% revenue share in 2025, while peripheral devices are projected to expand at a 7.61% CAGR through 2031.
- By application, medication administration held 39.55% of 2025 revenue, whereas diagnostics and testing is set to grow at a 7.68% CAGR to 2031.
- By material, polyurethane captured 48.25% share, whereas silicone is forecast to register an 7.85% CAGR over 2026-2031.
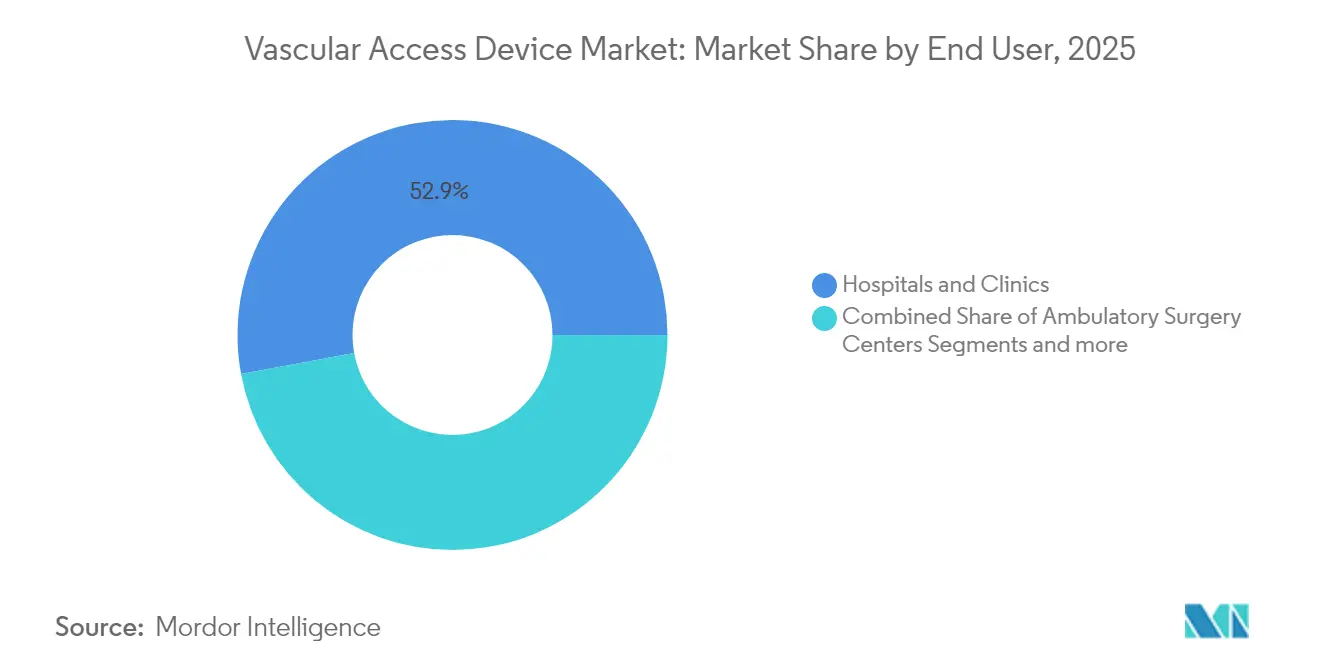
- By end user, hospitals and clinics accounted for 52.90% share in 2025, while ambulatory surgery centers (ASCs) are poised for an 7.77% CAGR.
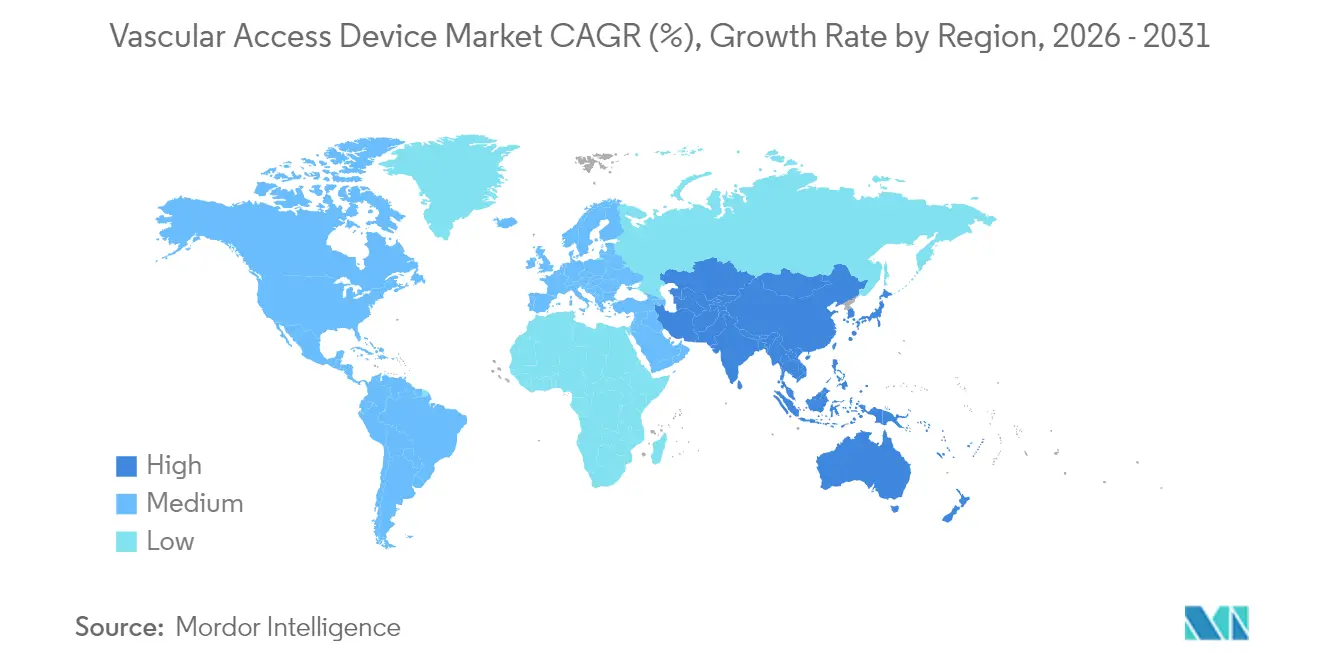
- By geography, North America dominated with 39.85% revenue share in 2025, but Asia-Pacific is projected to advance at an 7.95% CAGR.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Vascular Access Devices Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising burden of chronic diseases & high IV-therapy demand | 2.1% | Global, with concentration in North America & Europe | Long term (≥ 4 years) |
| Growth in chemotherapy procedures & hospitalization | 1.8% | Global, particularly developed markets | Medium term (2-4 years) |
| Increasing pediatric & neonatal vascular access | 1.2% | North America & Europe, expanding to APAC | Medium term (2-4 years) |
| Adoption of ultrasound-guided DIVA solutions | 1.5% | Global, led by North America | Short term (≤ 2 years) |
| Expansion of home & community infusion therapy | 1.9% | North America & Europe | Medium term (2-4 years) |
| Favorable reimbursement policies and guidelines | 0.8% | Primarily North America & Europe | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Rising burden of chronic diseases & high IV-therapy demand
Chronic conditions such as diabetes and heart failure are driving long-term intravenous treatment requirements worldwide. Home infusion now serves more than 3.2 million Americans annually, with spending exceeding USD 110 billion and rising 5-7% each year. Durable midline catheters and extended-dwell peripheral devices lower complication risk, supporting broader outpatient management. Hydrophilic biomaterials like Access Vascular’s MIMIX reduce failure rates and can save a 1,000-bed hospital USD 1.8 million a year. These economics elevate vascular access devices from commodity supplies to essential infrastructure.
Growth in chemotherapy procedures & hospitalization
Personalized oncology regimens increasingly depend on central venous catheters that tolerate vesicant drugs while permitting frequent sampling [1]Caitriona Duggan, "Vascular access devices for prolonged intravenous therapy regimens in people diagnosed with cancer," Cohrane Library, pmc.ncbi.nlm.nih.gov. Peripherally inserted central catheters (PICCs) improve outpatient flexibility, cutting treatment delays and hospital stays. Chlorhexidine-impregnated dressings have trimmed bloodstream infections by 52% in trials, underpinning premium pricing for infection-resistant devices [2]Huilin Xu, "Improving central venous catheter care with chlorhexidine gluconate dressings: evidence from a systematic review and Meta-analysis," BMC, jhpn.biomedcentral.com.
Increasing pediatric & neonatal vascular access
Neonatal intensive care units report 4-day average indwell times for extended-dwell peripheral IVs with 71.7% success, while PICCs reach 83.6%. The modified Seldinger technique minimizes vessel trauma in premature infants. Taurolidine locks cut bloodstream infections by 45% and hospitalizations by 41% in children on parenteral nutrition. Pediatric-specific R&D therefore remains a strategic differentiator.
Adoption of ultrasound-guided DIVA solutions
Difficult intravenous access affects up to 35% of surgical patients, costing U.S. emergency departments USD 2.68 billion annually. Ultrasound triples first-attempt success, yet two-thirds of clinicians still rely on blind cannulation. One-hand guidewire kits achieve 75% first-try success versus 50% for standard methods. Vendors pairing catheters with bedside imaging and training capture outsized mindshare [3]Amit Bahl, "An Improved Definition and SAFE Rule for Predicting Difficult Intravascular Access (DIVA) in Hospitalized Adults," Journal of Infusion Nursing, journals.lww.com.
Restraints Impact Analysis*
| Restraint | % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Catheter-associated bloodstream infections (CLABSI) | -1.4% | Global, particularly acute care settings | Medium term (2-4 years) |
| Stringent regulatory scrutiny & product recalls | -0.9% | Global, led by FDA and European regulators | Short term (≤ 2 years) |
| Alternate long-acting drug-delivery routes | -0.6% | Developed markets with advanced pharmaceutical infrastructure | Long term (≥ 4 years) |
| Medical-grade PU & silicone supply constraints | -1.1% | Global, with particular impact on Asia-Pacific manufacturing | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Catheter-associated bloodstream infections (CLABSI)
Intensive-care units still record 4.9 infections per 1,000 catheter days, with every episode adding treatment cost and mortality risk. Digital dashboards cut CLABSI rates by up to 73% but require capital investment. Chlorhexidine dressings reduce catheter colonization by 54%. Hospitals consequently favor integrated infection-control bundles, squeezing commodity vendors that lack advanced coatings.
Stringent regulatory scrutiny & product recalls
FDA alerts on catheter material fatigue and its harmonized Quality System Regulation demand costly redesigns that smaller firms struggle to meet. New 506J shortage-notification rules compel manufacturers to disclose supply disruptions six months ahead, exposing competitive information. The compliance burden accelerates consolidation in the vascular access devices market.
*Our forecasts treat driver/restraint impacts as directional, not additive. The impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Device Type: Central Dominance with Peripheral Momentum
Central devices captured 56.12% of vascular access devices market share in 2025. PICCs remain the workhorse for oncology and critical-care settings, whereas tunneled catheters support year-long therapies. Hospitals value their reliable flow and lower reinsertion frequency, even as reimbursement pressures intensify scrutiny of infection risk and dwell time.
Peripheral catheters, however, are the fastest-growing class at a 7.61% CAGR. Extended-dwell designs bridge the gap between standard PIVCs and PICCs, reducing cost and complication rates for intermediate therapies. FDA-cleared devices such as B. Braun’s Introcan Safety 2 extend median dwell to 5.7 days. Ultrasound-guided placement has broadened clinical acceptance, and antimicrobial polyurethane upgrades further differentiate offerings. As outpatient infusion volumes swell, peripheral innovations are positioned to gain additional vascular access devices market traction.

By Application: Therapeutic Delivery Leads, Diagnostics Accelerates
Medication administration commanded 39.55% of 2025 revenue, underlining the indispensable role of safe intravenous drug delivery. Complex biologics, chemotherapy cocktails and high-osmolar solutions necessitate robust central lines and ports capable of repeated access without integrity loss. Coated lumens and pressure-tolerant hubs have become standard, supporting price premiums.
Diagnostics and testing is forecast to grow at a 7.68% CAGR as precision-medicine protocols demand serial biomarker sampling. Point-of-care devices shorten turnaround times, spurring hospitals to favor catheters with low hemolysis rates and easy blood-draw access. Manufacturers integrating multi-lumen configurations and AI-optimized geometries offer tangible workflow savings, strengthening competitive positioning within the vascular access devices market.
By End User: Hospitals Retain Scale, ASCs Gain Velocity
Hospitals and clinics generated 52.90% of 2025 revenue because critical-care, emergency and surgical departments rely on high-performance vascular access platforms. Large networks negotiate bulk contracts that bundle catheters with integrated securement and antimicrobial accessories. Supply-chain shocks have led many systems to dual-source, allowing nimble suppliers to unseat incumbents.
ASCs represent the fastest-expanding channel with an 7.77% CAGR through 2031. Newly introduced C-codes lift reimbursement for cardiac catheterization by up to USD 3,346, validating same-day procedural economics. Private-equity backed ASC chains are scaling cardiology and vascular suites, elevating demand for user-friendly kits that speed turnover. Vendors re-engineering packaging for quick procedural setup are poised to capture incremental vascular access devices market sales.

By Material: Polyurethane Foundation, Silicone Upswing
Polyurethane held 48.25% share in 2025, reflecting decades of clinical familiarity and cost efficiency. Yet the material’s 30% complication rate in catheter cases drives hospitals to seek alternatives. Tight medical-grade PU supply following PTFE shortages has prompted larger firms to verticalize extrusion or lock in long-term contracts, buffering margin impact.
Silicone, advancing at an 7.85% CAGR, benefits from superior biocompatibility that lowers thrombosis. Surface-modified silicone infused with antimicrobial agents slashes bacterial adhesion and meets long-term implant needs. Pediatric specialists increasingly specify silicone lines for fragile vessels, making the material a strategic focus area. Hybrid constructs—combining silicone flexibility with polyurethane strength—are emerging, reinforcing material innovation as a core battleground in the vascular access devices market.
Geography Analysis
North America commanded 39.85% of 2025 revenue on the back of sophisticated healthcare infrastructure and an insurance mix that rewards infection-reduction technologies. BD invested more than USD 10 million in 2024 to expand U.S. catheter production, adding hundreds of millions of units annually and reinforcing domestic supply resilience. Terumo earmarked USD 30 million for Angio-Seal capacity in Puerto Rico, underscoring the region’s manufacturing pull.
Asia-Pacific is projected to post an 7.95% CAGR through 2031, reflecting rising chronic disease prevalence and healthcare spending. China’s shift to value-based procurement is exerting price pressure, yet local champions eye export markets to offset domestic margin compression. Japan-headquartered Terumo reported that its Rika mid-clamp platform is now installed in 98 centers across the region, nearing its 100-site milestone.
Europe maintains a robust installed base driven by strict infection-prevention mandates and early HTA adoption. Meanwhile, Middle Eastern health-system build-outs and South American economic recovery create pockets of high growth, especially where public insurers support outpatient infusion. Ongoing geopolitical frictions and raw-material constraints are encouraging firms to develop multi-hub sourcing models so the vascular access devices market can meet varied regional demand without disruption.
Competitive Landscape
The vascular access devices market shows moderate fragmentation, with multinationals leveraging broad portfolios, infection-control IP and global distribution. BD, Teleflex and ICU Medical anchor the top tier, while mid-sized firms differentiate through niche clinical focus or advanced material science. Infection-resistant coatings remain the centerpiece of premium positioning; heparin network surface treatments have cut thrombus formation by 62.5% in preclinical work.
Technology convergence is quickening. ICU Medical and Otsuka formed a USD 200 million venture that lifts annual IV-solution capacity to 1.4 billion units and embeds closed-loop safety valves. Robotic insertion and AI-guided selection tools, though nascent, are beginning to influence procurement criteria, especially among teaching hospitals.
Portfolio rationalization continues. AngioDynamics divested its PICC and midline brands to Spectrum Vascular for USD 45 million to focus on thrombus management platforms. Supply-chain reliability is growing in strategic importance; manufacturers able to assure redundancy in resin sourcing and sterilization capacity enjoy preferential contract status. As outpatient settings expand, design priorities tilt toward ease-of-use and rapid turnover, fostering a steady pipeline of incremental innovations that keep competition brisk across the vascular access devices market.
Vascular Access Devices Industry Leaders
-
NIPRO Medical Corporation
-
B. Braun Melsungen AG
-
Baxter International Inc.
-
Becton, Dickinson and Company
-
Teleflex Inc.
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- February 2025: Teleflex Incorporated announced the acquisition of BIOTRONIK’s Vascular Intervention business for approximately EUR 760 million (USD 820 million), broadening its interventional cardiology offering.
- May 2025: BD revealed plans to separate its Biosciences and Diagnostic Solutions unit, creating a USD 17.8 billion MedTech entity that includes its vascular access franchise.
- January 2025: Penumbra launched the Element Vascular Access System, the first laser-cut hypotube sheath compatible with Lightning Flash 2.0 for venous thromboembolism treatment.
- October 2024: BD introduced the BD Intraosseous Vascular Access System for emergency settings where conventional IV placement is delayed.
Research Methodology Framework and Report Scope
Market Definitions and Key Coverage
Our study defines the vascular access device (VAD) market as the global sale of purpose-built catheters and ports that create short- or long-term venous entry for drug infusion, fluid or nutrition delivery, blood sampling, hemodialysis, and hemodynamic monitoring. Device families covered include central venous catheters, implantable ports, peripherally inserted central catheters, midlines, short peripheral IV catheters, butterfly needles, and related securement accessories.
Scope Exclusions: diagnostic guidewires, vascular closure devices, and peripheral stents are excluded.
Segmentation Overview
-
By Device Type
-
Central Vascular Access Devices
- Peripherally-Inserted Central Catheters (PICCs)
- Non-tunnelled Catheters
- Tunnelled Catheters
- Other Central Vascular Access Devices
-
Peripheral Vascular Access Devices
- Peripheral IV Catheters (PIVC)
- Midline Catheters
- Other Peripheral Vascular Access Devices
-
Central Vascular Access Devices
-
By Application
- Medication or Drug Administration
- Fluid and Nutrition Administration
- Blood and Blood-Product Transfusion
- Diagnostics and Testing
- Other Applications
-
By End User
- Hospitals & Clinics
- Ambulatory Surgery Centers
- Others
-
By Material
- Polyurethane
- Silicone
- Others
-
By Geography
-
North America
- United States
- Canada
- Mexico
-
Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Rest of Europe
-
Asia-Pacific
- China
- Japan
- India
- Australia
- South Korea
- Rest of Asia-Pacific
-
Middle East and Africa
- GCC
- South Africa
- Rest of Middle East and Africa
-
South America
- Brazil
- Argentina
- Rest of South America
-
North America
Detailed Research Methodology and Data Validation
Primary Research
Interviews with vascular surgeons, infusion nurses, supply-chain managers, and regional distributors across North America, Europe, and Asia-Pacific helped us validate adoption curves, typical catheter mix, and price pass-throughs. Short web surveys with nephrologists clarified chronic-dialysis access replacement rates and regional practice preference shifts.
Desk Research
We began with authoritative public datasets, including FDA 510(k) clearances, EUDAMED registrations, and HCPCS reimbursement volumes, because they reveal annual device introductions and baseline procedural counts. Trade bodies such as the Infusion Nurses Society, Kidney Disease Outcomes Quality Initiative, and Global Dialysis Network supply guideline changes and dialysis prevalence that shape demand. Hospital purchasing disclosures, select 10-K filings, and peer-reviewed infection-rate studies further anchor average selling prices and dwell-time assumptions. Subscription resources, notably D&B Hoovers for company revenues and Dow Jones Factiva for shipment-linked news, round out financial signals. This list is illustrative; many additional open and paid sources informed data checks.
Market-Sizing & Forecasting
A top-down build starts with hospital and home-care infusion procedures, dialysis sessions, and oncology infusion courses; these volumes are multiplied by verified device-per-procedure ratios and blended regional ASPs. Bottom-up cross-checks, including supplier revenue roll-ups and sampled tender data, fine-tune totals before final alignment.
Key model drivers include: - annual chemotherapy patient counts - chronic dialysis population growth - infection-related replacement incidence - average catheter dwell time - regulatory shifts affecting power-injectable port uptake.
Forecasts to 2030 use multivariate regression with dialysis prevalence, oncology incidence, and health-expenditure growth as predictors; scenario analysis adjusts for large-scale supply-chain disruptions. Data gaps in emerging markets are bridged with calibrated regional proxies and expert-confirmed penetration rates.
Data Validation & Update Cycle
Outputs pass three-layer variance checks, senior analyst review, and anomaly reconciliation. Models refresh each year, with interim updates triggered by material recalls, reimbursement changes, or macro shocks. A final pre-publication sweep ensures the figures clients receive are the most current view.
Why Our Vascular Access Device Baseline Earns Exceptional Trust
Published estimates often differ; definitions, product mix, and refresh cadence rarely match.
Key gap drivers include whether intraosseous devices are counted, how catheter accessories are priced, and the frequency of model updates. Mordor's analysts revisit regulatory filings annually and apply blended ASPs derived from procurement audits, whereas many firms rely on static list prices or merge vascular closure and peripheral devices into one pool.
Benchmark comparison
| Market Size | Anonymized source | Primary gap driver |
|---|---|---|
| USD 6.25 B (2025) | Mordor Intelligence | - |
| USD 5.99 B (2024) | Global Consultancy A | excludes IO devices; updates bi-annually; pure top-down build |
| USD 8.64 B (2024) | Trade Journal B | combines closure and peripheral devices; uses undisclosed ASP inflation |
These comparisons show that our disciplined scope selection, dual-track validation, and annual refresh provide a balanced, transparent baseline that decision-makers can replicate and trust.


Key Questions Answered in the Report
What is the current vascular access devices market size and its projected growth rate?
The market is valued at USD 6.68 billion in 2026 and is forecast to grow at a 6.92% CAGR, reaching USD 9.34 billion by 2031.
Which device category commands the largest share and which is expanding the fastest?
Central vascular access devices lead with a 56.12% share in 2025, while peripheral devices post the highest growth at a 7.61% CAGR through 2031.
Which region contributes the most revenue, and which shows the strongest growth momentum?
North America accounts for 39.85% of global revenue in 2025, whereas Asia-Pacific is set to advance at an 7.95% CAGR.
What primary driver has the greatest positive influence on market expansion?
The rising burden of chronic diseases and high IV-therapy demand adds an estimated +2.1% to the overall CAGR.
How do hospitals compare with ambulatory surgery centers (ASCs) in market performance?
Hospitals and clinics hold 52.90% market share in 2025, whereas ASCs are the fastest-growing channel at an 7.77% CAGR.
Which materials dominate, and which material segment is growing quickest?
Polyurethane maintains a 48.25% share, while silicone is the fastest-growing material with an 7.85% CAGR.
Page last updated on:









