United States Active Pharmaceutical Ingredients (API) Market Analysis by Mordor Intelligence
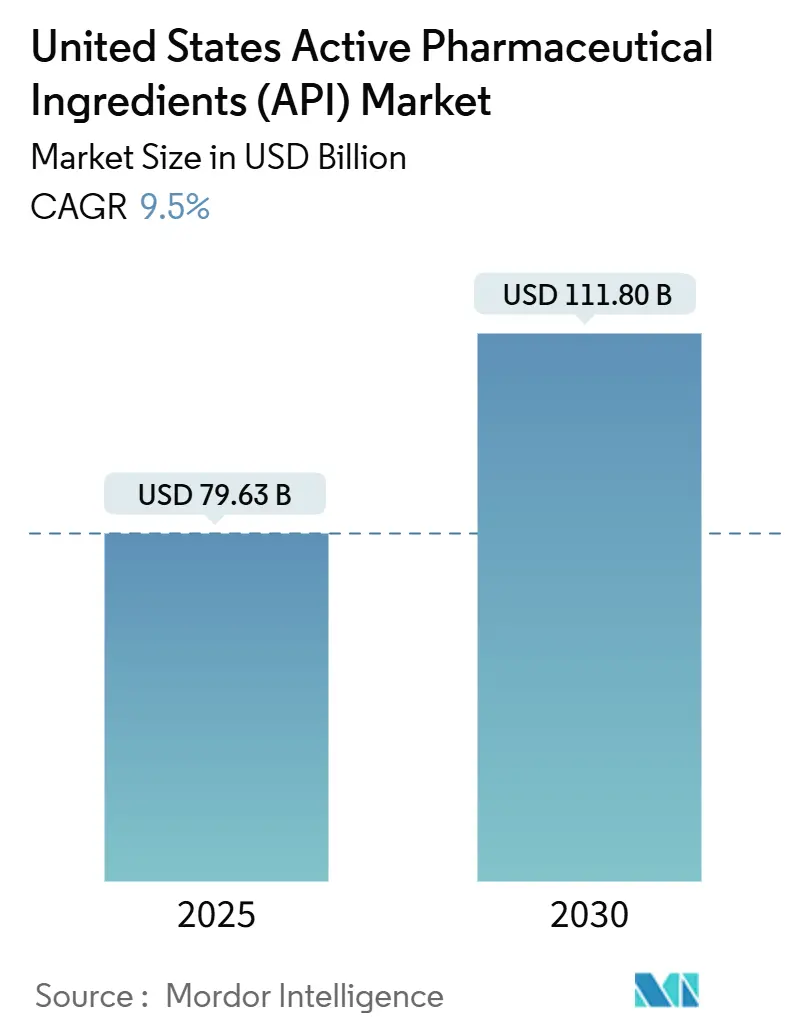
The United States Active Pharmaceutical Ingredients market size stood at USD 79.63 billion in 2025 and is projected to expand at a 6.91% CAGR, reaching USD 111.80 billion by 2030. Supply-chain fragilities exposed by COVID-19 and escalating geopolitical risk have prompted more than USD 160 billion in pledged Big Pharma manufacturing investments since 2020. mRNA breakthroughs and a broader biologics pipeline are intensifying demand for complex large-molecule ingredients, while oncology’s pivot toward high-potency compounds reallocates capital toward specialized containment facilities. Continuous manufacturing and AI-enabled quality systems are shortening cycle times and mitigating Medicare price pressures through productivity gains. Strategic clustering in North Carolina, Indiana, and Michigan compresses lead times by colocating skilled labor, regulators, and logistics nodes.
Key Report Takeaways
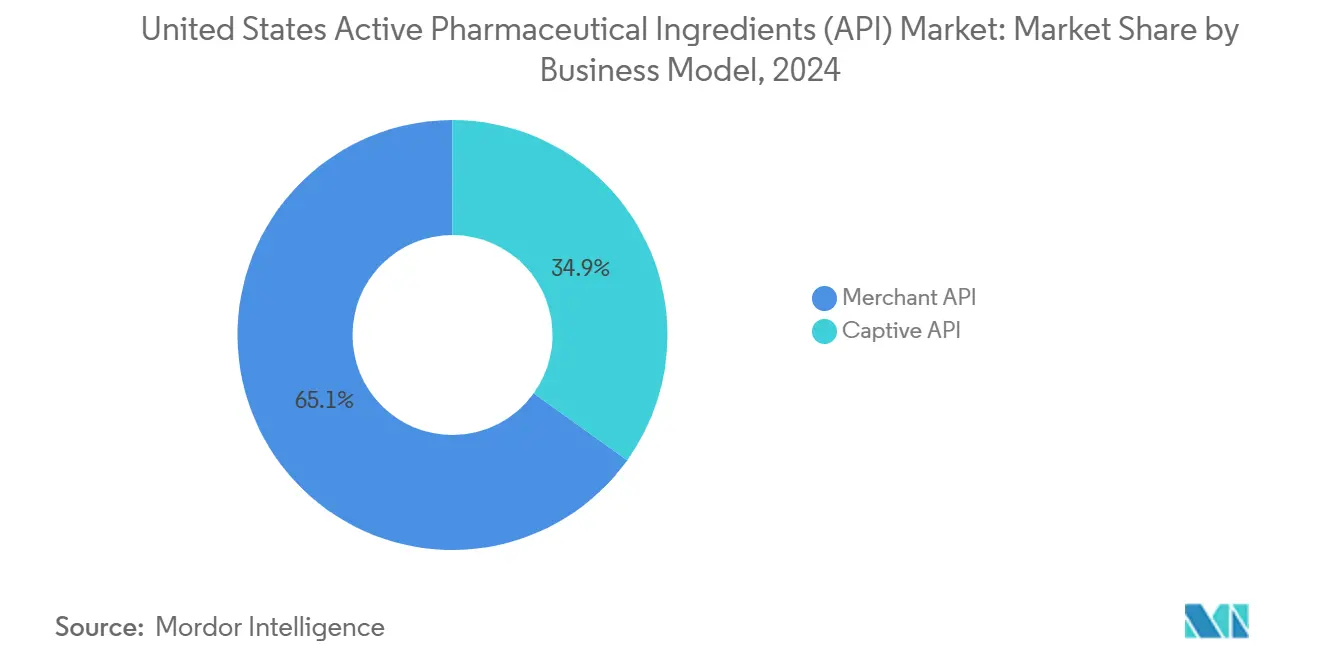
By business mode, merchant API operations commanded 65.09% United States Active Pharmaceutical Ingredients market share in 2024 and are expanding at a 7.19% CAGR through 2030.
By synthesis type, synthetic APIs retained 71.35% share in 2024, whereas biotech APIs are advancing at a 7.33% CAGR to 2030.
By molecule size, small molecules held 63.50% share in 2024, yet large molecules are forecast to post the fastest 7.47% CAGR through 2030.
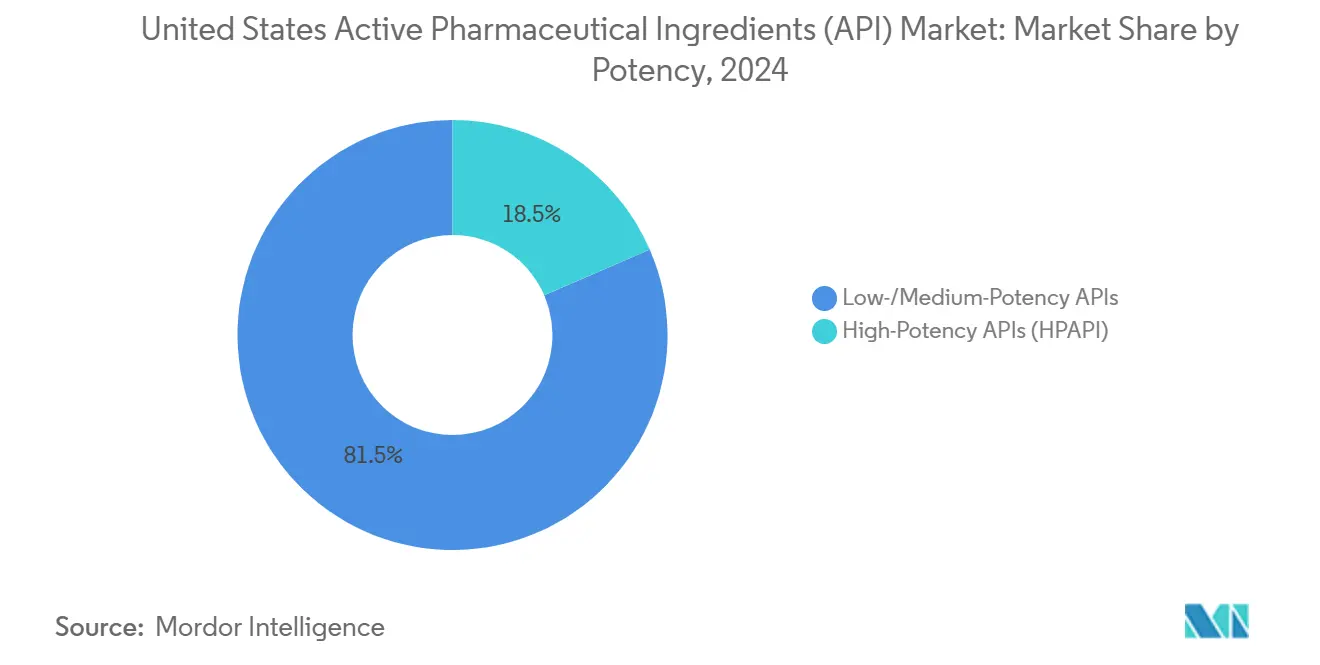
By potency, low- and medium-potency APIs represented 76.50% of the United States Active Pharmaceutical Ingredients market size in 2024, while high-potency APIs are growing at 7.62% CAGR.
By application, oncology led with 28.71% share in 2024 and is projected to grow at a 7.77% CAGR to 2030.
United States Active Pharmaceutical Ingredients (API) Market Trends and Insights
Driver Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Diversifying U.S. supply-chain incentives for domestic API manufacturing | +1.8% | National; North Carolina, Indiana, Michigan | Medium term (2-4 years) |
| Surge in advanced biologics & mRNA therapy pipelines | +1.5% | Research Triangle, Boston, San Francisco | Long term (≥ 4 years) |
| Federal funding for pandemic-preparedness stockpiles | +1.2% | Nationwide strategic-reserve sites | Short term (≤ 2 years) |
| Growing demand for high-potency APIs (HPAPIs) from oncology boom | +1.4% | Michigan, North Carolina, Kentucky | Medium term (2-4 years) |
| Adoption of continuous manufacturing technologies | +0.9% | Established pharma hubs nationwide | Long term (≥ 4 years) |
| Contract development & manufacturing outsourcing surge among virtual pharma | +1.1% | CDMO-rich regions across the U.S. | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Diversifying U.S. Supply-Chain Incentives for Domestic API Manufacturing
A USD 34 billion federal program, coupled with Eli Lilly’s USD 27 billion factory build-out, has reset the cost calculus for on-shore API production. Incentives such as tax credits and accelerated depreciation shrink China’s historical 35-40% cost edge. The API Innovation Center is tasked with reshoring 25% of small-molecule supply within five years under a USD 14 million grant.
Surge in Advanced Biologics & mRNA Therapy Pipelines
Biologics CDMOs are expanding 9-11% a year, outpacing small-molecule growth because personalized medicine needs sophisticated fermentation and lipid-system capabilities. Fujifilm’s USD 1.2 billion North Carolina upgrade adds 160,000 L of bioreactor capacity. Croda secured USD 75 million in government backing to expand U.S. lipid production for mRNA therapies. AI-assisted workflows are boosting biologics throughput by 20%.
Federal Funding for Pandemic-Preparedness Stockpiles
A USD 105 million HHS allocation pivots policy from reactive import procurement toward pre-emptive U.S. production of sterile injectables and critical APIs. Contracts linked to the Strategic National Stockpile extend 7-10 years, giving manufacturers revenue certainty unavailable in commercial channels. Continuous-manufacturing adopters enjoy further incentives under FDA’s Advanced Manufacturing Technologies Designation Program, shaving months off review cycles and rewarding early movers[1]Source: FDA, “Advanced Manufacturing Technologies Designation Program,” fda.gov . This public-private model de-risks large-scale domestic capacity, narrowing cost gaps with overseas rivals while elevating national resilience.
Growing Demand for High-Potency APIs from the Oncology Boom
Targeted cancer therapies require sub-microgram containment, pushing manufacturers toward specialized clean-room suites, barrier isolators, and occupational-exposure limits far stricter than those for traditional APIs. Consequently, HPAPI margins remain resilient even as Medicare negotiations compress pricing on commodity molecules [2]Source: Centers for Medicare & Medicaid Services, “Medicare Drug Price Negotiation,” cms.gov . Piramal’s USD 80 million Lexington upgrade and BeiGene’s USD 800 million New Jersey site underscore how players are doubling down on potency-focused infrastructure that few newcomers can match . The FDA imposes intensified inspection protocols on HPAPI facilities, which, while capital-intensive, create durable competitive moats.
Restraint Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Price erosion from Medicare Drug Price Negotiations | –1.6% | Nationwide | Short term (≤ 2 years) |
| Environmental compliance costs for U.S. API plants | –0.8% | States with stricter regulations | Medium term (2-4 years) |
| Shortage of skilled process chemists & engineers | –1.2% | Traditional manufacturing regions | Long term (≥ 4 years) |
| Rising competition from Indian & Chinese low-cost producers | –1.4% | Global; impacts U.S. export competitiveness | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Price Erosion from Medicare Drug Price Negotiations
Initial CMS talks slashed prices by 22-80% for ten high-volume drugs, stripping USD 6 billion from annual sales in a single round. The 2025 list broadens to 15 products, four of them oncology agents, setting a precedent for across-the-board cost resets that ripple directly to upstream API suppliers. Because negotiations group all indications sharing an active ingredient, manufacturers must peg API costs to the lowest-margin use case, squeezing profitability unless they pivot toward biosimilars and generics exempt from the rules.
Shortage of Skilled Process Chemists & Engineers
Eighty percent of U.S. plants struggle to fill advanced process roles, and retirements compound the gap just as digital twins, PAT, and continuous reactors become standard . Universities are revising curricula, but a four-to-five-year lag leaves current expansion projects competing fiercely for a limited talent pool, delaying ramp-ups and inflating labor costs. CDMOs with internal training academies obtain a head start, suggesting that workforce strategy, not capital, could become the decisive growth constraint.
Segment Analysis
By Business Mode: Merchant Dominance Drives Outsourcing
Merchant providers captured 65.09% of the United States Active Pharmaceutical Ingredients market in 2024, and their 7.19% CAGR through 2030 far outpaces captive operations. CDMOs are evolving into CRDMOs that knit discovery, CMC, and commercial supply into one workflow, trimming development timelines by 50%. Virtual pharmaceutical firms without bricks-and-mortar plants represent the fastest-growing client block, driving small-volume, high-complexity orders that favor merchant flexibility. Continuous-manufacturing expertise lets these providers swing between batches in hours instead of days, a capability captive units rarely match.
Strategic alliances reinforce merchant leadership: Recipharm’s pact with Exela opens a North Carolina sterile line able to run 100 million units annually. Merchant plants also gain FDA fast-track benefits for innovative equipment, shortening validation cycles. Several Big Pharma companies are spinning surplus reactors into merchant subsidiaries, boosting asset utilization while preserving quality oversight. Heightened due-diligence standards after past contamination events create a reputational moat favoring established CDMOs. As Medicare negotiations lower margins on commoditized drugs, innovators lean on merchants to squeeze out 5-10% extra efficiency that captive networks often cannot unlock.
By Synthesis Type: Biotech Acceleration Reshapes Production
Synthetic chemistry still delivered 71.35% of 2024 revenue, yet biotech APIs are climbing at 7.33% CAGR as personalized medicine scales. Viral-vector, mRNA, and recombinant-protein workloads demand stainless-steel and single-use bioreactors, driving capex per cubic meter far above small-molecule norms. Firms that once specialized in solvent-based synthesis now pursue hybrid footprints through M&A or joint ventures.
Continuous bioprocessing lets hybrid plants exploit real-time analytics common to flow-chemistry lines, smoothing the learning curve. Upstream convergence shows in PAT sensors that monitor both cell viability and solvent purity, reducing training hours for operators. Still, each 200,000 L mammalian suite costs roughly USD 800 million, so first movers enjoy scale economics that late entrants struggle to replicate. Regulatory harmonization between synthetic and biologic modules remains incomplete, forcing dual audit tracks that raise compliance spend by 12-15% but also slow copy-cat capacity abroad.
By Molecule Size: Large-Molecule Innovation Drives Premium Growth
Small molecules claimed 63.50% share in 2024, yet large molecules are advancing at 7.47% CAGR on the back of monoclonal antibodies and gene therapies. Average selling prices per gram run 20-30-times higher than for oral solids, letting biologic batches recoup capex faster. Hybrid antibody–drug conjugates require both chem-linker know-how and protein expertise, nudging manufacturers to co-locate capabilities.
Large-molecule fill-finish relies on cold-chain networks able to hold −80 °C stock, prompting partnerships with 3PL providers that specialize in biologics. FDA inspection loads are heavier, yet accelerated approval pathways for orphan drugs offset the burden. Small-molecule incumbents responding through micro-flow reactors shave solvent use 30%, narrowing cost gaps but not eliminating biologic momentum. Investors continue to reward biologic exposure with valuation multiples 1.5-2× higher than for pure small-molecule peers, sustaining capital inflow.
By Potency: HPAPI Premium Positioning Counters Pricing Pressure
Low- and medium-potency classes covered 76.50% of 2024 volume, yet HPAPIs are growing 7.62% a year and command premium margins. Containment suites built to Occupational Exposure Level 5 add USD 3,000–4,000 per square foot, quadrupling spend versus standard blocks but delivering three-to-four-times higher gross profit. Medicare price caps largely spare niche oncology launches, bolstering HPAPI revenue predictability.
EPA air-emission limits under 40 CFR 63 Subpart GGG require carbon-adsorption or cryogenic-condensation units, costs that deter under-capitalized entrants. Workforce hurdles intensify; certified industrial hygienists and containment engineers remain scarce, allowing firms with established talent to dictate timelines. Some generic houses are pooling resources for shared HPAPI hubs to spread fixed costs, but technology-transfer complexity prolongs validation and keeps incumbents ahead.
By Application: Oncology Leadership Drives Value Creation
Oncology held 28.71% share in 2024 and is racing ahead at 7.77% CAGR as immunotherapies, targeted degraders, and radioligand therapies move through trials. Each drug class needs unique API sub-components, from linker toxins to alpha emitters, deepening specialization. High-potency containment boosts per-batch profit, cushioning producers against Medicare negotiations that erode commodity margins. Emerging antibody–drug conjugates further blur molecule-size boundaries by combining small cytotoxics with large antibodies in single constructs.
Cardiology, pulmonology, ophthalmology, neurology, and orthopedic indications together supply volume but less value, often subject to sharper price controls. Cardiology APIs may regain momentum once GLP-1 obesity therapies demonstrate cardio-protective endpoints, nudging suppliers to repurpose peptide lines. Neurology segments benefit from breakthrough-therapy designations in Alzheimer’s and depression, though biologic blood-brain-barrier delivery remains an unresolved hurdle. Application diversity thus mitigates demand swings, but oncology will dictate capex priorities through 2030.
By End User: Outsourced Partners and Hospital Networks Anchor Off-Take
Contract development and manufacturing organizations (CDMOs) remain the dominant end-user group because virtual pharmaceutical companies and legacy brand owners alike rely on external specialists for complex synthesis, sterile fill–finish, and high-potency containment needs. Merchant suppliers therefore channel most capacity expansions toward CDMOs that bundle discovery, clinical-trial material, and commercial volumes under single quality systems, minimizing tech-transfer risk for innovators. These healthcare providers prioritize APIs sourced from U.S. facilities with continuous-manufacturing lines because shorter lead times translate into fewer back-orders—a consideration magnified by recent shortages of sterile injectables.
The rise of precision oncology and orphan therapies further realigns end-user mix: specialty biopharma firms without bricks-and-mortar assets now account for a growing share of small-batch, high-potency API contracts, pulling production toward CDMOs that invest in glove-box isolators and single-use suites. Together, CDMOs, healthcare systems, and public stockpiles shape an end-user landscape that rewards flexibility, rapid scale-up capability, and compliant domestic footprints—traits that align closely with the ongoing re-shoring trajectory of the United States Active Pharmaceutical Ingredients market
Geography Analysis
North Carolina anchors domestic production, drawing more than USD 15 billion from Eli Lilly, Amgen, and Novo Nordisk in just three years. The Research Triangle’s proximity to three Tier-1 universities and two international airports compresses tech-transfer and export lead times. Nearly 34,000 residents work across 108 biomanufacturing sites, giving companies a ready labor base reinforced by NIIMBL training stipends. State grants covering 2–3% of capex sweeten project IRRs, while Duke Energy’s below-national-average industrial rates shave ongoing overhead. NC’s “plug-and-play” shell-building program further accelerates green-field builds, cutting up-front construction time by six months and supporting rapid scale-ups for pandemic surge capacity.
Indiana’s USD 9 billion Lebanon complex becomes the nation’s largest synthetic-API site and benefits from an established petrochemical feed-stock corridor. The Midwest location taps competitive electricity and water tariffs, narrowing operating-expense gaps with Asia. Michigan focuses on HPAPI production, leveraging automotive-sector precision-machining know-how for glove-box fabrication. Ohio’s injectables cluster around Columbus exploits FedEx and UPS ground hubs, enabling 1-day delivery to 60% of U.S. hospitals. Illinois and Wisconsin contribute secondary suppliers of sterile filters, single-use bags, and silicone tubing, reinforcing a Midwest self-sufficiency loop that reduces inter-state freight miles.
Missouri’s API Innovation Center, backed by USD 9.55 million in state support, illustrates how smaller states can seed new clusters and dilute geographic-concentration risk. California and Washington still house the bulk of discovery labs yet remain short on late-stage API capacity, prompting discussions around co-located “lab-to-line” campuses. Texas markets its low-tax stance and Gulf Coast chemical backbone to lure future synthetic-API lines, though water-scarcity concerns could temper momentum. Puerto Rico, after historic tax-credit losses, is courting biologic fill-finish to revive its once-dominant footprint. A diversified map thus emerges: coastal R&D clusters feed mid-Atlantic and Midwest production hubs, while interior states use incentives to capture spillover as capacity keeps re-shoring.
Competitive Landscape
The top five suppliers hold roughly half of the revenue, indicating moderate concentration. Pfizer, Merck, and Johnson & Johnson retain end-to-end capabilities yet outsource niche chemistries to Cambrex or Thermo Fisher. Patent-expiration exposure of USD 183.5 billion fuels consolidation and tech-driven productivity plays. AI-enabled process controls trimmed Pfizer’s deviation rate 30% and cycle time 20%. CordenPharma’s USD 981 million GLP-1 peptide push signals cross-Atlantic rivalry for obesity-therapy supply.
United States Active Pharmaceutical Ingredients (API) Industry Leaders
-
Pfizer Inc.
-
Novartis AG
-
BASF SE
-
Viatris Inc.
-
Teva Pharmaceutical Industries Ltd
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- February 2025: Eli Lilly pledged more than USD 50 billion for new U.S. manufacturing, including three API sites
- December 2024: Amgen committed USD 1 billion for a second Holly Springs plant
- October 2024: Lilly unveiled the USD 4.5 billion Lilly Medicine Foundry
United States Active Pharmaceutical Ingredients (API) Market Report Scope
An Active Pharmaceutical Ingredient (API) is a part of any drug that produces its effects. Some drugs, such as combination therapies, have multiple active ingredients to treat different symptoms or act in different ways. They are produced using highly technological industrial processes during research and development and the commercial production phase.
The United States Active Pharmaceutical Ingredients (API) Market is Segmented by Business Mode (Captive API and Merchant API), Synthesis Type (Synthetic and Biotech), Drug Type (Generic and Branded), and Application (Cardiology, Oncology, Pulmonology, Neurology, Orthopedic, Ophthalmology, and Other Applications). The report offers the value (in USD billion) for the above segments.
| Captive API |
| Merchant API |
| Synthetic |
| Biotech |
| Small-Molecule |
| Large-Molecule / Biologics |
| High-Potency APIs (HPAPI) |
| Low/Medium-Potency APIs |
| Cardiology |
| Pulmonology |
| Oncology |
| Ophthalmology |
| Neurology |
| Orthopedic |
| Other Applications |
| Domestic Pharma Manufacturers |
| Multinational Pharma Subsidiaries (KSA) |
| CDMOs / CMOs |
| Hospitals & Research Institutes |
| By Business Mode | Captive API |
| Merchant API | |
| By Synthesis Type | Synthetic |
| Biotech | |
| By Molecule Size | Small-Molecule |
| Large-Molecule / Biologics | |
| By Potency | High-Potency APIs (HPAPI) |
| Low/Medium-Potency APIs | |
| By Application | Cardiology |
| Pulmonology | |
| Oncology | |
| Ophthalmology | |
| Neurology | |
| Orthopedic | |
| Other Applications | |
| By End-User | Domestic Pharma Manufacturers |
| Multinational Pharma Subsidiaries (KSA) | |
| CDMOs / CMOs | |
| Hospitals & Research Institutes |


Key Questions Answered in the Report
How fast is domestic API production growing in the United States Active Pharmaceutical Ingredients market?
Value is rising at a 6.91% CAGR between 2025 and 2030, supported by federal incentives and over USD 160 billion in private investment.
Which synthesis route is expanding quickest?
Biotech APIs are advancing at a 7.33% CAGR thanks to mRNA and biologics pipelines.
What share do merchant providers hold?
Merchant operations captured 65.09% share in 2024 and continue to outpace captive models.
Why is North Carolina so prominent?
The Research Triangle offers skilled talent, regulatory proximity, and USD 15 billion-plus in recent plant commitments, making it the leading cluster.
How will Medicare price negotiations affect suppliers?
Initial rounds cut prices up to 80%, pressuring traditional API margins but creating openings in biosimilars and HPAPIs exempt from negotiation.
Page last updated on:







