Market Trends of Global Thrombectomy Devices Industry
This section covers the major market trends shaping the Thrombectomy Devices Market according to our research experts:
The Mechanical Thrombectomy Devices Segment is the largest segment of the thrombectomy devices market to witness the highest growth over the forecast period.
Mechanical thrombectomy refers to a new technique of stroke treatment that eliminates blood clots from the brain's major blood arteries. The operation is advised for individuals who have had an acute ischemic stroke caused by a major arterial obstruction in the brain's anterior circulation. Growing preference for the method is likely to augur well for the global mechanical thrombectomy market shortly.
However, with a rise in the burden of ischemic strokes across the globe, a novel treatment option known as mechanical thrombectomy has been made available by medical device companies. For instance, in 2021 NeuroVasv announced the ENVI-SR Mechanical Thrombectomy System (ENVI-SR), a stent-retriever being developed by NeuroVasc Technologies for the removal of blood clots in patients with acute ischemic stroke due to large vessel occlusion. For another instance, in July 2021, Surmodics, a leading provider of medical devices and in vitro diagnostic technologies to the health care industry, announced the acquisition of privately held Vetex Medical Limited. The Galway, Ireland-based medical device developer and manufacturer has focused exclusively on venous clot removal solutions. The transaction expands Surmodics' thrombectomy portfolio with a second United States Food and Drug Administration (USFDA) 510(k)-cleared device, the ReVene Thrombectomy Catheter. The ReVene mechanical thrombectomy catheter is specifically designed to remove large, mixed-morphology blood clots commonly found with venous thromboembolism (VTE). The device's dual action technology efficiently removes mixed-morphology clots in a single session, minimizing the need for thrombolytics and without capital equipment. Due to these technological advancements, the mechanical thrombectomy device segment is expected to be the largest segment, and witness to show significant growth over the forecast period.
Mechanical thrombectomy significantly reduces stroke-related disabilities in patients when used in conjunction with tPA and other medical treatments. The rise in demand for minimally invasive surgical procedures and favorable reimbursement policies for cardiovascular and neurovascular procedures are expected to boost demand for mechanical thrombectomy devices during the forecast period.
North America Expected to Dominate the Market Over the Forecast Period
The increasing burden of cardiac patients, technological advancements in the devices to attain better clinical outcomes, and increasing demand for minimally invasive products are driving the thrombectomy, presence of key market players driving the thrombectomy devices market in North America.
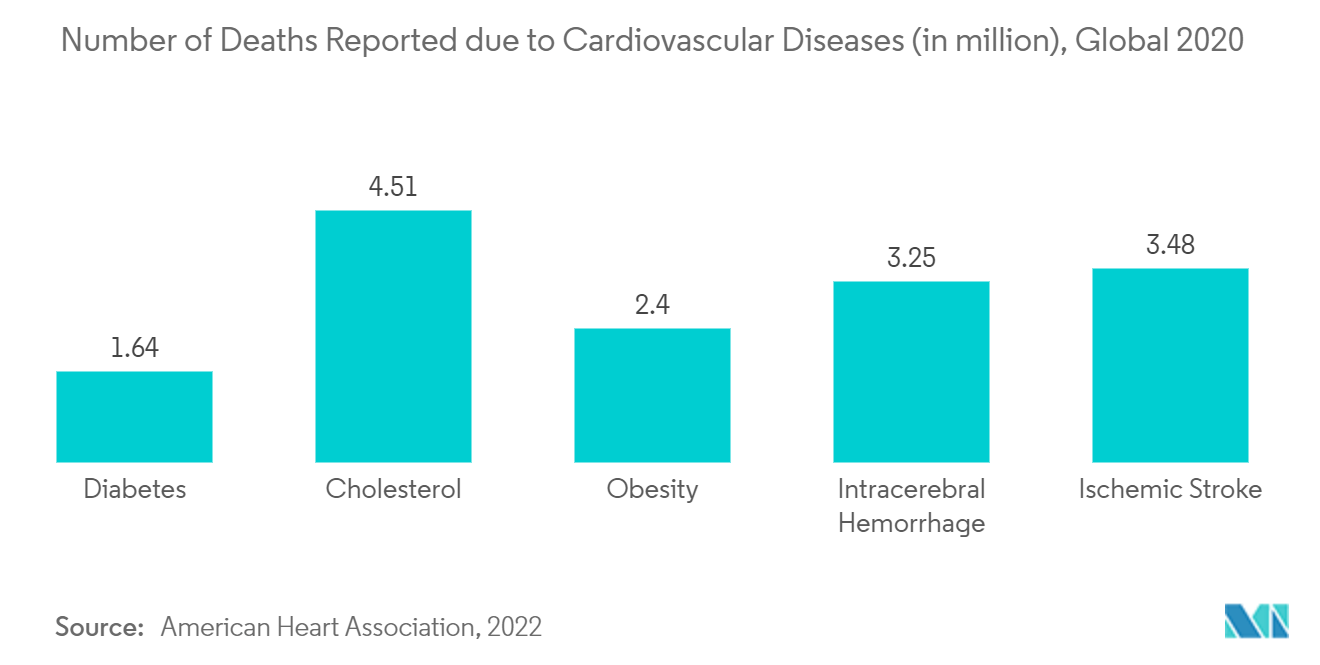
The rising cases of cardiac patients, suffering from heart attack and cardiac arrest are increasing in the North American region, which is mainly attributed due their lifestyles and diet pattern another reason for the increasing burden of cardiac patients in that region. For instance, in 2022 Centre for Disease Control and Prevention (CDC) reported that 805,000 people had a heart attack in the United States. Of these 605,000 are a first heart attack and 200,000 happen to people who have already had a heart attack. For another instance, in 2022 an article published in the National Library of Medicine reported that in the United States, about 610,000 people die of heart disease every year. That is 1 of every 4 deaths. Coronary heart disease is the leading cause of death in the Western world killing over 370,000 people annually. On average, about 735,000 Americans have a heart attack every year. Thus the high burden of cardiac diseases in the region drives the thrombectomy devices market.
Key market players are also involved in marketing strategies like a merger, acquisitions, etc in the segment tracked to gain a competitive edge in the region. For instance, in September 2021, Abbott announced the acquisition of Walk Vascular, LLC, a commercial-stage medical device company with a minimally invasive mechanical aspiration thrombectomy system designed to remove peripheral blood clots. For another instnace, in August 2021, Stryker Neurovascular received the regulatory United States Food and Drug Administration (USFDA) approval for their Trevo NXT Pro Vue Retriever, a class II medical device classified as a neurovascular mechanical thrombectomy device for acute ischemic stroke. Such acquistitions and advancement is driving the market in North America.
Therefore, due to the favorable reimbursement scenario for medical devices and aforementioned factors, the market is estimated to grow consistently in the North American region.