Sweden In-Vitro Diagnostics Market Analysis
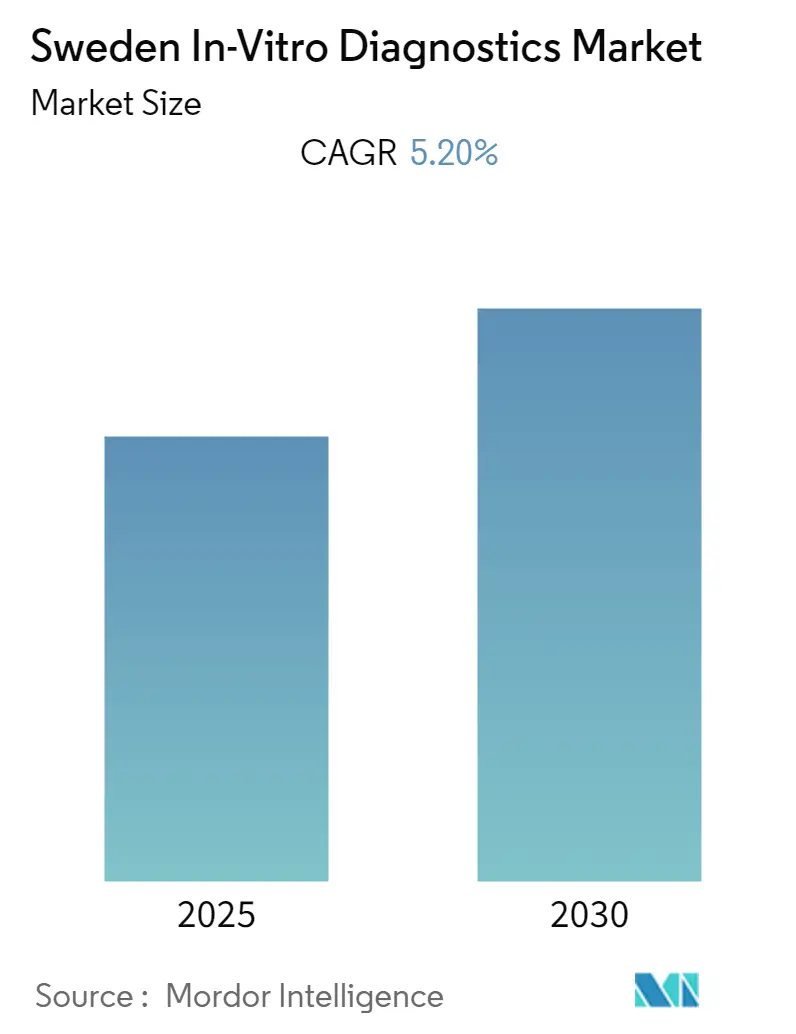
The Sweden In-Vitro Diagnostics Market is expected to register a CAGR of 5.2% during the forecast period.
- Certain factors that are driving the market growth include high prevalence of chronic diseases, increasing use of point-of-care (POC) diagnostics, advanced technologies, and increasing awareness and acceptance of personalized medicine and companion diagnostics.
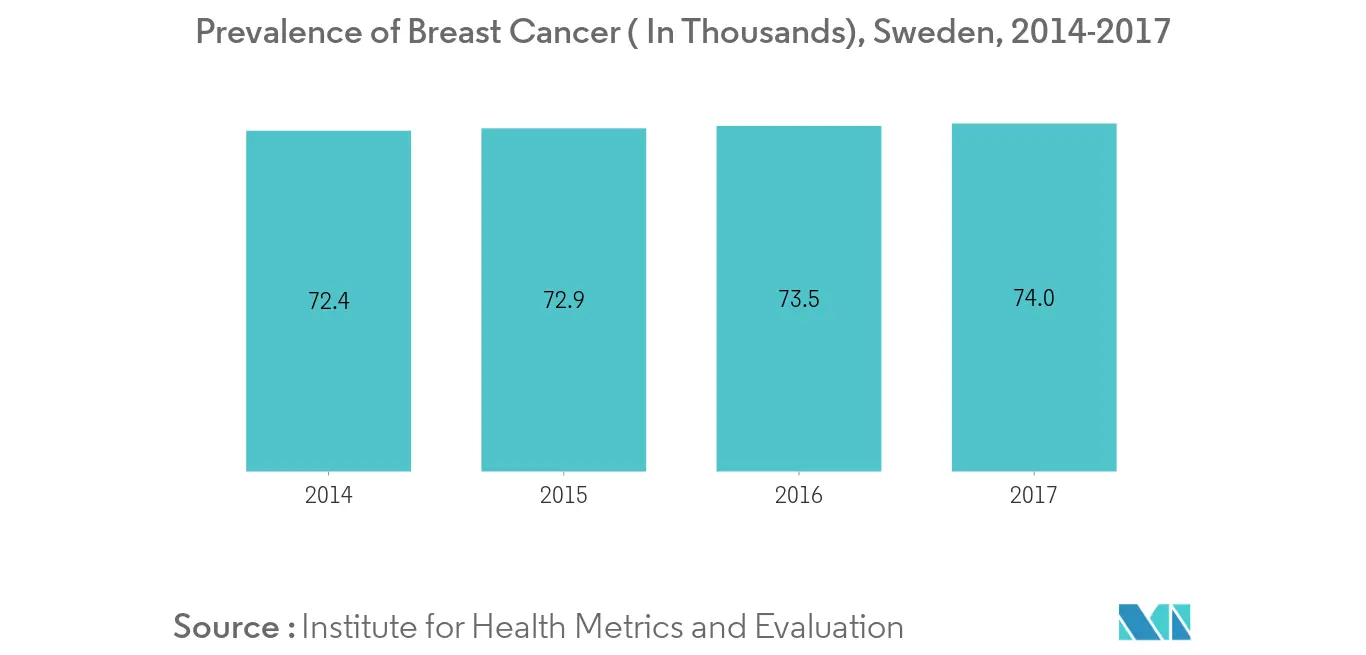
- Rising geriatric population is expected to be one of the major factor impacting growth of the market. According to Statistics Sweden's population projection, in 2018, the share of people aged 80 and older will increase most to 19.86% of total population, and in 2028, their share is expected to be 255,000 more people than today, an increase of 50 percent.
- Effective management of patients with one or more chronic disease is a commonly debated concern in Sweden. More effective primary care delivery may increase the overall efficiency and responsiveness of the health system. Hence, the increasing health expenditure in the country is expected to fuel the market growth over the forecast period.
Sweden In-Vitro Diagnostics Market Trends
The Reagent Segment is Expected to Hold the Highest Market Share During the Forecast Period
The reagent segment of the market studied includes chemical, biological, or immunological components, solutions, or preparations intended by the manufacturer to be used during the in vitro diagnosis process. Given the high cost of many diagnostic platforms, it is common for manufacturers to lease equipment instead of selling technology outright to end users. In these arrangements, the lease is tied to contracts to purchase associated reagents or assays for the equipment over the term of the contract. Many diagnostic companies have in excess of 75% of sales from consumables, such as assays and reagents, and such agreements guarantee the generation of cyclic revenues associated with the sale of reagents and other consumables.
Sweden In-Vitro Diagnostics Industry Overview
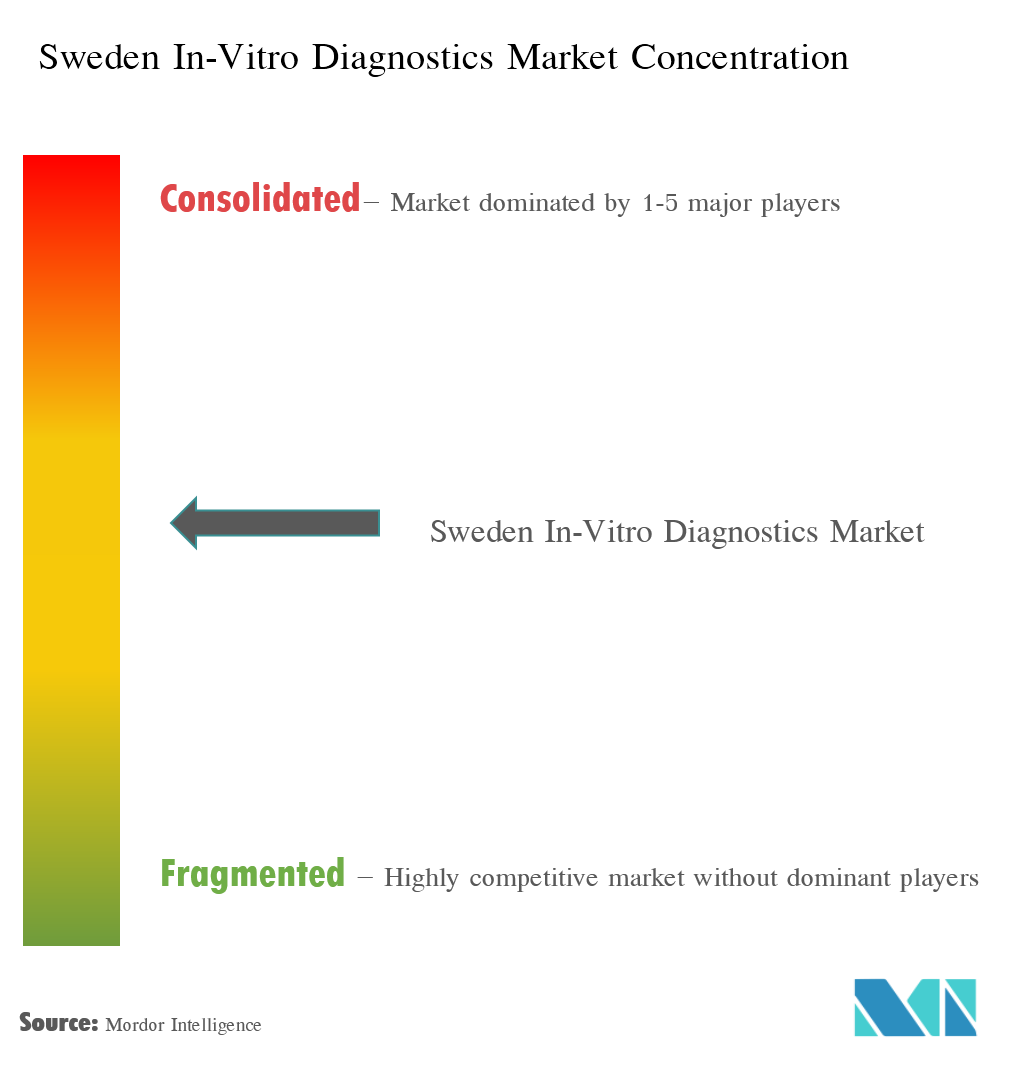
The Swedish in vitro diagnostics market is highly competitive and consists of a number of major players. Companies, like Abbott Laboratories, Becton, Dickinson, and Company, Bio-Rad Laboratories Inc., Danaher Corporation, Johnson & Johnson, Roche Diagnostics, Siemens Healthcare, and Thermo Fisher Scientific Inc., among others, hold the substantial market share in the Swedish in vitro diagnostics market.
Sweden In-Vitro Diagnostics Market Leaders
-
Abbott Laboratories
-
Becton, Dickinson, and Company
-
Bio-Rad Laboratories, Inc.
-
Danaher Corporation
-
Thermo Fisher Scientific Inc.
- *Disclaimer: Major Players sorted in no particular order
Sweden In-Vitro Diagnostics Industry Segmentation
As per the scope of this report, in vitro diagnostics involves medical devices and consumables that are utilized to perform in vitro tests on various biological samples. They are used for the diagnosis of various medical conditions, such as chronic diseases.
| By Test Type | Clinical Chemistry |
| Molecular Diagnostics | |
| Immuno Diagnostics | |
| Haematology | |
| Other Types | |
| Product | Instrument |
| Reagent | |
| Other Products | |
| Usability | Disposable IVD Devices |
| Reusable IVD Devices | |
| Application | Infectious Disease |
| Diabetes | |
| Cancer/Oncology | |
| Cardiology | |
| Autoimmune Disease | |
| Other Applications | |
| End Users | Diagnostic Laboratories |
| Hospitals & Clinics | |
| Other End Users |
Sweden In-Vitro Diagnostics Market Research FAQs
What is the current Sweden In-Vitro Diagnostics Market size?
The Sweden In-Vitro Diagnostics Market is projected to register a CAGR of 5.2% during the forecast period (2025-2030)
Who are the key players in Sweden In-Vitro Diagnostics Market?
Abbott Laboratories, Becton, Dickinson, and Company, Bio-Rad Laboratories, Inc., Danaher Corporation and Thermo Fisher Scientific Inc. are the major companies operating in the Sweden In-Vitro Diagnostics Market.
What years does this Sweden In-Vitro Diagnostics Market cover?
The report covers the Sweden In-Vitro Diagnostics Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Sweden In-Vitro Diagnostics Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Our Best Selling Reports
France Ophthalmic Devices Industry Report
Statistics for the 2025 Sweden In-Vitro Diagnostics market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Sweden In-Vitro Diagnostics analysis includes a market forecast outlook for 2025 to 2030 and historical overview. Get a sample of this industry analysis as a free report PDF download.