Global Spinal Fusion Devices Market Analysis by Mordor Intelligence
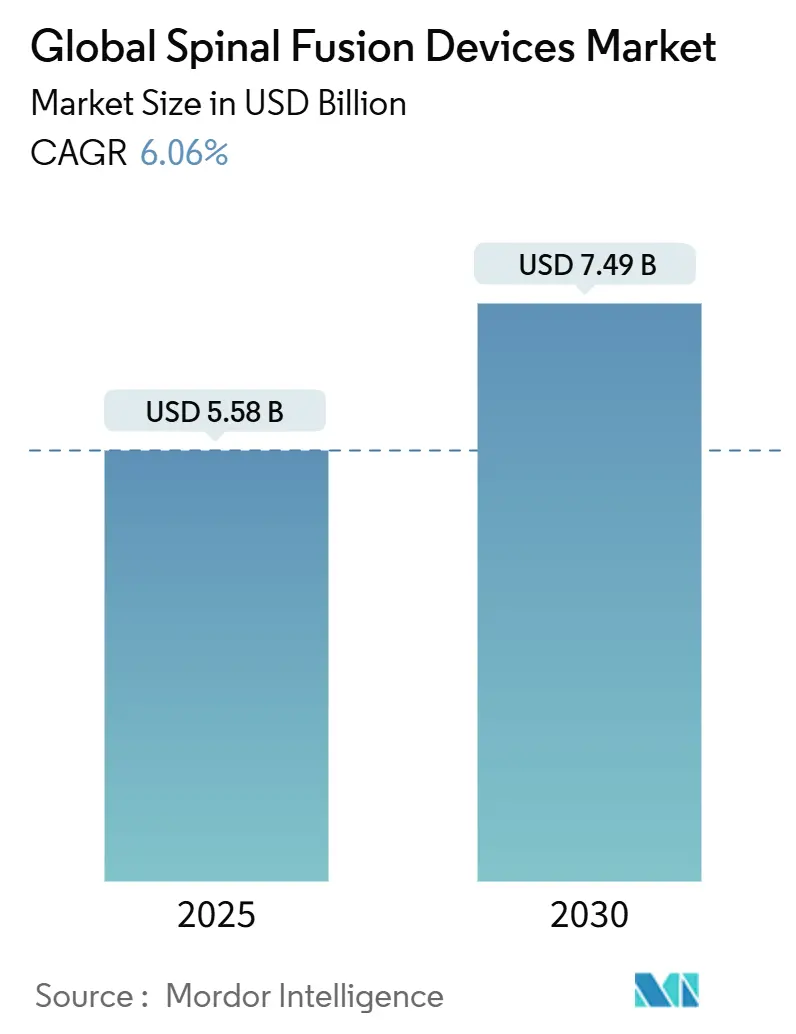
The spinal fusion devices market size is estimated at USD 5.58 billion in 2025 and is projected to climb to USD 7.49 billion by 2030, delivering a 6.06% CAGR. Robust demand stems from demographic aging, rising degenerative spine disorders, and accelerating adoption of minimally invasive and AI-enabled surgical platforms. Hospitals and ambulatory surgery centers (ASCs) are scaling procedure volumes as Medicare has more than quadrupled the number of fusion procedures reimbursed in outpatient settings, reinforcing steady case-mix migration. Rapid FDA clearances for 3-D-printed patient-specific cages and the 96.99% screw-placement accuracy achieved by contemporary robotic systems are intensifying competitive differentiation. In parallel, payers’ shift toward bundled payments is pressuring pricing but is also catalyzing the development of value-driven implants, stimulating technological innovation across every product class.
Key Report Takeaways
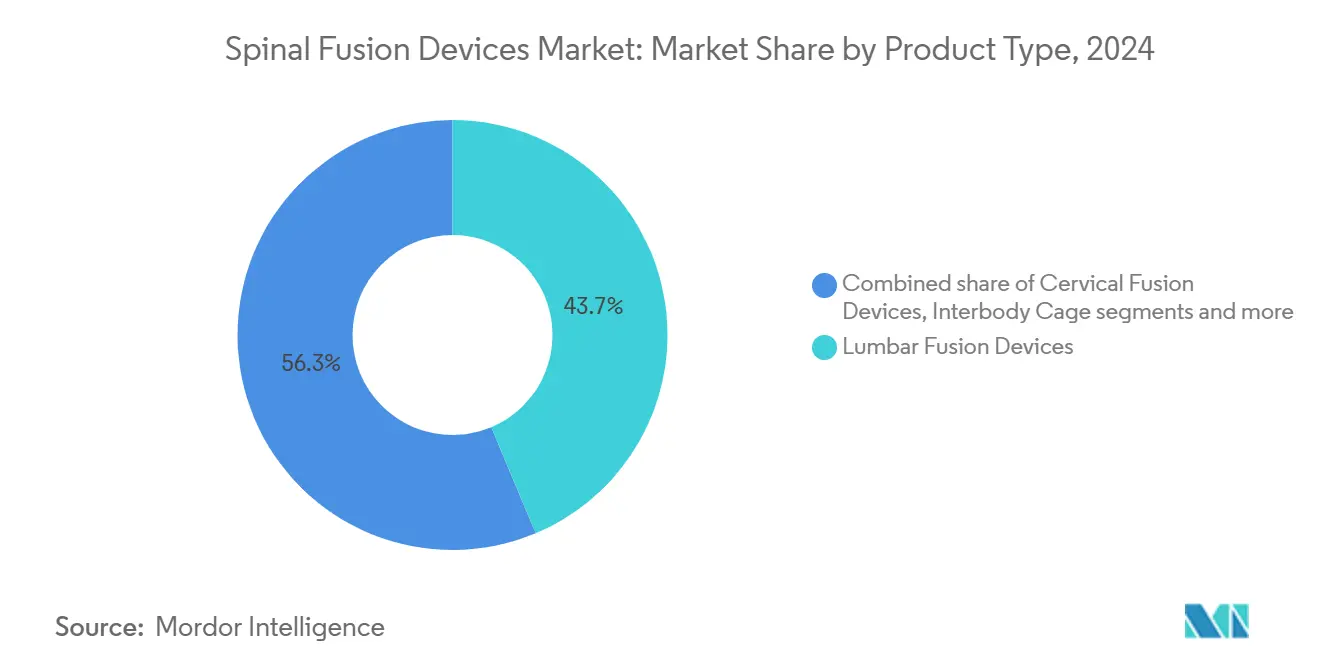
- By product type, lumbar fusion devices led with 43.68% of spinal fusion devices market share in 2024, while interbody cages are on track for a 7.02% CAGR through 2030.
- By surgery type, minimally invasive procedures captured 62.37% revenue in 2024; open surgery is trailing but remains essential for complex deformities. The minimally invasive segment is expanding at 6.34% CAGR to 2030.
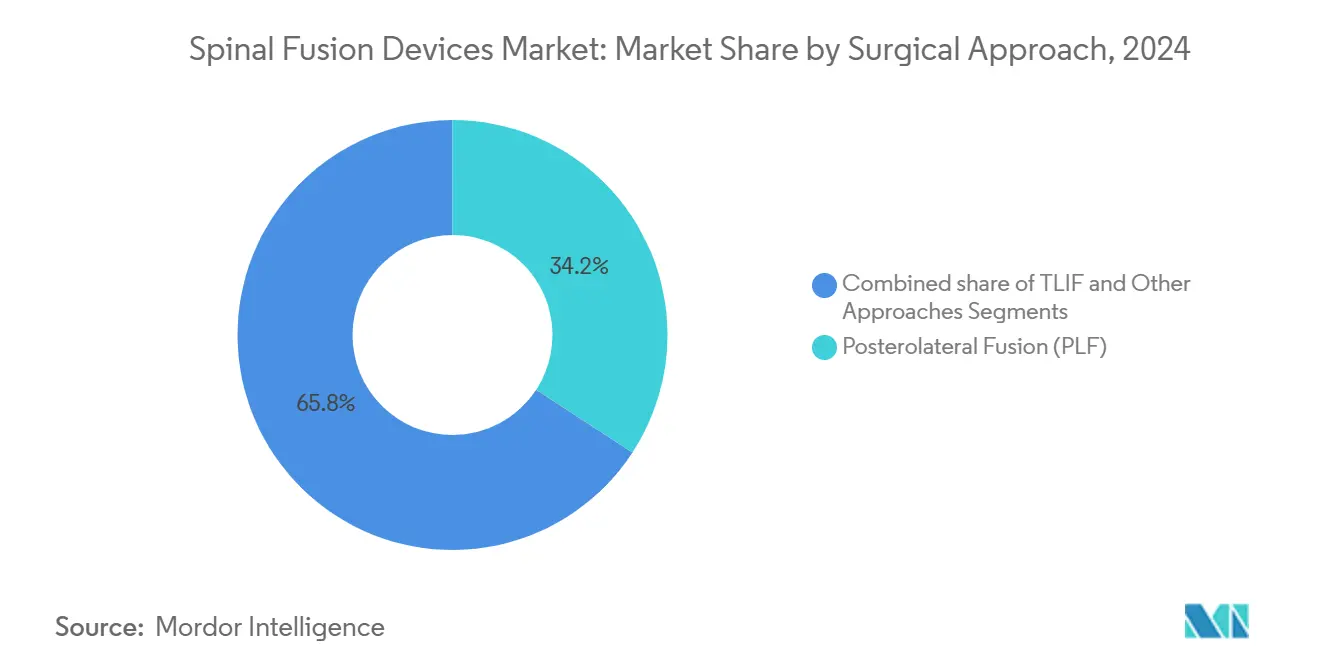
- By surgical approach, transforaminal lumbar interbody fusion (TLIF) recorded the fastest growth at 6.61% CAGR, whereas posterolateral fusion still held 34.21% revenue in 2024.
- By end-user, hospitals accounted for a 62.54% share of the spinal fusion devices market size in 2024, yet ASCs are set to accelerate at 6.78% CAGR to 2030.
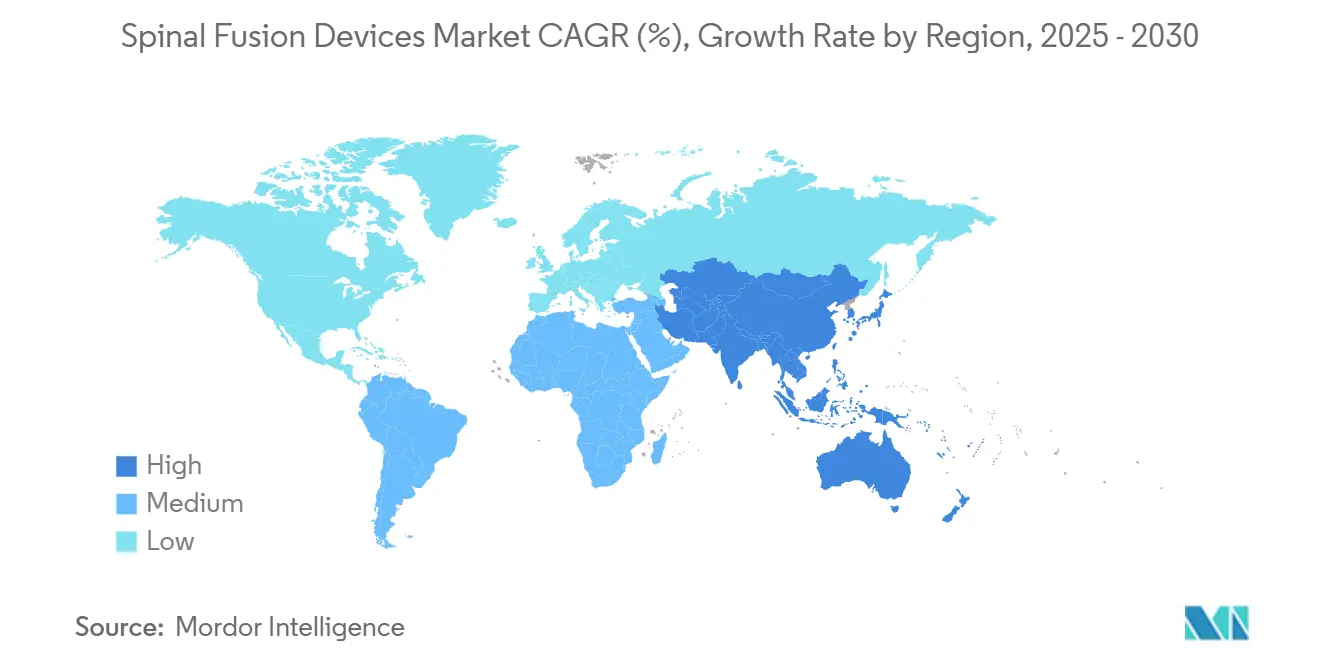
- By region, North America retained 46.23% of the global total in 2024; Asia–Pacific is the fastest-growing geography at a 6.95% CAGR.
Global Spinal Fusion Devices Market Trends and Insights
Drivers Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Minimally invasive fusion techniques | +1.8% | North America & Europe leading, Asia-Pacific following | Medium term (2–4 years) |
| Prevalence of degenerative spine disorders | +1.5% | Global, concentrated in developed markets | Medium term (2–4 years) |
| Expanding geriatric population | +1.2% | Global, highest in North America & Europe | Long term (≥ 4 years) |
| Wider outpatient reimbursement | +0.9% | North America primary, expanding to Europe | Short term (≤ 2 years) |
| AI-guided robotic navigation | +0.7% | North America & Europe early, Asia-Pacific emerging | Medium term (2–4 years) |
| 3-D-printed patient-specific cages | +0.5% | Global, innovation led by North America & Europe | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Shift toward minimally invasive fusion techniques
Minimally invasive spine surgery is overturning long-standing open procedures by delivering shorter hospital stays[1]Zhaojun Song, “Short-term and mid-term evaluation of three types of minimally invasive lumbar fusion surgery for treatment of L4/L5 degenerative spondylolisthesis,” Scientific Reports, nature.com , smaller incisions, and lower complication rates while maintaining equal fusion success. Meta-analyses of transforaminal lumbar interbody fusion confirm fewer transfusions and a 4.83% complication rate versus 14.97% for open surgery. Robotic navigation drives screw accuracy beyond 96%, prompting device manufacturers to bundle implants, navigation, and intra-operative imaging. Fellowship programs now prioritize these techniques, ensuring a pipeline of surgeons fluent in robot-assisted workflows. Hospitals also leverage the faster patient recovery to improve bed turnover, directly aligning clinical performance with value-based purchasing.
Escalating prevalence of degenerative spine disorders
Sedentary lifestyles, obesity, and greater diagnostic scrutiny have raised lumbar disc degeneration incidence to more than 90% in individuals past 60 years. Earlier imaging enables timely surgical referral, averting progression to multilevel disease. Health-economic analyses prove that early fusion lowers chronic pain expenditure, and payers are expanding coverage accordingly. Clinical registries confirm that early single-level lumbar fusion reduces re-operation frequency and boosts quality-adjusted life years. Device makers respond by broadening portfolios of expandable cages and biologics optimized for single-level pathology.
Growing geriatric population & life-expectancy gains
By 2050, the global 65+ cohort will double, fostering steady fusion demand. Medicare reports a 193% growth in spine procedures from 2010–2021, including a 15.7% annual jump at ASCs. Older recipients experience similar fusion rates when minimally invasive techniques limit blood loss. Implant suppliers increasingly tailor instrumentation to osteoporotic bone, while biologic developers explore osteo-enhancing grafts. The demographic tide guarantees a durable patient base even if reimbursement pressure tightens.
Increasing reimbursement coverage for outpatient spine procedures
Medicare’s outpatient list now covers 58 fusion-related codes versus 12 in 2010, saving USD 140 million annually on anterior cervical fusions. Complication rates average below 2% in ASC settings, convincing private payers to roll out bundled payments that reward efficient care. Device vendors specifically design single-use, sterile-packed kits to fit ASC economics. Ambulatory centers reciprocate by installing compact CT scanners and image-guided towers, further propelling the spinal fusion devices market.
Restraints Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High implant costs vs. value-based care | –1.1% | North America & Europe primary, emerging in Asia-Pac | Short term (≤ 2 years) |
| Adjacent-segment disease scrutiny | –0.9% | Global, highest in developed markets | Medium term (2–4 years) |
| Stringent FDA & MDR approval timelines | –0.8% | North America & Europe | Medium term (2–4 years) |
| Surgeon shortage in emerging economies | –0.6% | Asia-Pacific, Latin America, MEA | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
High implant costs vs. value-based-care payment models
Bundled-payment pilots cap total episode cost, forcing providers to weigh implant performance against price. Cervical fusion intra-operative expenses reach USD 7,574, with 69% tied to hardware. Hospitals renegotiate volume contracts, favoring platforms that minimize re-operation liability. Manufacturers now issue evidence dossiers[2]Timothy J. Yee, “Cost-Effectiveness of Posterior or Transforaminal Lumbar Interbody Fusion for Grade 1 Lumbar Spondylolisthesis: A 5-Year Quality Outcomes Database Study,” Journal of Neurosurgery: Spine, thejns.org showing cost per quality-adjusted life year below USD 100,000 to defend premium tags. Firms unable to articulate value risk share erosion in the spinal fusion devices market.
Rising scrutiny on adjacent-segment disease outcomes
Long-term follow-ups reveal 23.6% revision rates at 10 years, spurring interest in motion-preserving alternatives. Surgeons incorporate prophylactic alignment strategies and hybrid constructs to blunt adjacent-segment loading. Device makers invest in dynamic stabilization and nucleus augmentation technologies. Persistent payer inquiries into lifetime effectiveness could temper uptake of certain rigid constructs.
Segment Analysis
By Product Type: Lumbar Devices Drive Volume Growth
Lumbar fusion devices generated 43.68% of the 2024 spinal fusion devices market share, cementing their role as workhorse solutions for spondylolisthesis and disc degeneration. The spinal fusion devices market size for lumbar instrumentation is projected to expand at a 5.8% CAGR as demand persists across both inpatient and outpatient channels. Interbody cages stand out with a 7.02% growth rate thanks to 3-D-printed titanium lattices that secure 97% fusion success[3]Anna-Katharina Calek, “3D-Printed Titanium Cages for Anterior and Lateral Lumbar Interbody Fusion Result in Excellent One-Year Fusion Rates,” Global Spine Journal, journals.sagepub.com . Surgeons favor expandable cages that restore disc height and sagittal balance without excessive nerve retraction. Cervical plates and screws maintain consistent uptake for anterior cervical discectomy and fusion, underscored by their long safety record. Thoracic systems meet niche trauma and deformity needs but are turning to modular constructs for inventory efficiency. Pedicle screw innovation now focuses on navigated insertion and torque-limiting drivers, reducing mal-position. Biologic graft substitutes, including cellular bone allografts, realize 98.5% fusion, narrowing reliance on iliac crest autografts.
Continued material science advances exploit porous PEEK and magnesium alloys that encourage osteointegration while dampening stress shielding. Patient-specific implants, produced in days, personalize endplate coverage and load-sharing characteristics. Vendors increasingly package cage-graft bundles, simplifying logistics for ASCs. Still, value-analysis committees scrutinize unit price, steering hospitals toward platforms demonstrating both clinical superiority and cost-effectiveness, a balance that will define winners across the spinal fusion devices market.
Note: Segment shares of all individual segments available upon report purchase
By Type of Surgery: Minimally Invasive Techniques Reshape Practice
Minimally invasive procedures held 62.37% spinal fusion devices market size in 2024, posting a 6.34% CAGR through 2030 as imaging, navigation, and tubular retractors converge to curtail tissue disruption. Open surgery retains a role in severe deformity corrections yet faces shrinking share as robotic guidance[4]Jin-Jye Wan, “Robot-Assisted Minimally Invasive Transforaminal Interbody Fusion: A Complete Workflow Pilot Feasibility Study,” Journal of Spine Surgery, jss.amegroups.org abbreviates learning curves. Real-time 3-D imaging permits percutaneous pedicle screw trajectories with sub-2 mm deviation, lessening neurologic risk. Meanwhile, single-position spine techniques limit patient flips, shaving anesthesia time. Hospitals harness these efficiencies to qualify more cases for outpatient discharge, buoying ASC adoption and reshaping reimbursement dynamics.
The spinal fusion devices market responds with compact instrument suites, sterile-packed implants, and disposable navigation arrays tailored to ASC throughput. Training centers augment cadaveric labs with mixed-reality simulators, accelerating surgeon competency. Payers reward minimally invasive pathways via bundled reimbursement uplift for low complication incidence. As evidence matures, regulators may green-light shorter clearance paths for kits demonstrably improving safety, further embedding minimally invasive approaches into mainstream spine care.
By Surgical Approach: TLIF Emerges as Preferred Technique
Posterolateral fusion accounted for 34.21% of spinal fusion devices market share in 2024 but cedes momentum to TLIF, which is rising at a 6.61% CAGR as biomechanical advantages gain clinical validation. TLIF enables circumferential fusion from a unilateral corridor, preserving contralateral musculature and cutting intra-operative blood loss by up to 50% versus traditional bilateral facetectomy. Robot-assisted, minimally invasive TLIF enhances cage placement accuracy, promoting superior lordosis restoration and disc height maintenance. The spinal fusion devices market size for TLIF-compatible cages and screws is forecast to outpace legacy posterior systems through 2030.
Alternative routes such as anterior lumbar interbody fusion (ALIF) thrive in cases demanding maximal sagittal correction, while lateral approaches minimize dorsal muscle trauma. Surgeons increasingly rely on patient-specific planning to decide between TLIF, ALIF, or lateral strategies. Finite-element models published in 2024 detail stress profiles for each approach, informing implant geometry that mitigates adjacent-segment overload. Ongoing refinement will likely harmonize approach selection with personalized risk assessment.
Note: Segment shares of all individual segments available upon report purchase
By End-User: ASCs Capture Growing Market Share
Hospitals preserved 62.54% of the spinal fusion devices market size in 2024 as they manage polytrauma and complex revisions, yet the ASC channel is expanding at 6.78% CAGR, fueled by Medicare’s outpatient coverage expansion. ASCs operate lean staffing ratios and rely on fast-turnover implants, compelling suppliers to streamline tray counts and offer consignment options. Clinical series report sub-2% complication rates for one- to two-level lumbar fusions in ASC environments. Payers leverage that safety record to contract bundled payments featuring gain-sharing clauses.
Hospitals respond with hospital-outpatient departments that mimic ASC efficiency while retaining access to critical care back-up. Specialty orthopedic clinics, though not primary surgical sites, influence implant selection through pre-operative planning and long-term follow-up data. The spinal fusion devices market will see sustained channel diversification as technology further reduces length of stay and enables same-day discharge for multi-level constructs.
Geography Analysis
North America contributed 46.23% to global revenue in 2024 and should grow at 5.37% CAGR through 2030 as premium robotics and 3-D-printed implants penetrate both inpatient and outpatient settings. Medicare’s 58 ASC-eligible spine codes have catalyzed a 15.7% annual procedure rise in ambulatory centers, underpinning the spinal fusion devices market trend toward lower-cost sites of care. FDA breakthrough designations expedite commercialization, reinforcing the region’s innovation leadership.
Asia–Pacific is the swiftest climber with a 6.95% CAGR to 2030, leveraging hospital infrastructure upgrades, rising disposable incomes, and an expanding base of fellowship-trained spine surgeons. China’s Class III registration requirements are lengthy, yet local partnerships ease market access, and provincial tenders often favor cost-effective, yet technologically advanced, domestically produced implants. Japan and South Korea add volume through aging demographics, while India’s private hospital chains import navigated systems to capture medical tourism.
Europe’s 5.80% CAGR reflects balanced growth moderated by MDR compliance costs that slightly slow new-product launches. National health services encourage outpatient migration, particularly in the Nordics and Germany, but pricing controls challenge premium device margins. South America advances at 6.12% CAGR as Brazil and Argentina upgrade tertiary centers and adopt minimally invasive techniques. Middle East and Africa post 6.46% CAGR on the back of Gulf States’ specialist hospital investments, although surgeon shortages restrain broader regional uptake. Overall, the spinal fusion devices market is expanding worldwide, yet growth vectors differ markedly by reimbursement climate, surgeon density, and regulatory velocity.
Competitive Landscape
The spinal fusion devices market is moderately consolidated: the top five suppliers control an estimated 62% of global sales, while a vibrant cadre of start-ups targets performance niches. Globus Medical’s USD 250 million purchase of Nevro augments motion-preservation and pain-management synergies, signaling convergence between hardware and neuromodulation. Stryker’s divestiture of legacy spinal implants, while retaining its Mako Spine robot, highlights a pivot toward software-driven ecosystems. Medtronic’s AiBLE platform integrates navigation, imaging, and analytics, contributing high single-digit segment growth and locking surgeons into proprietary workflows.
Emerging players exploit additive manufacturing to deliver patient-specific cages; Curiteva logged 2,000 implants without revision in its first post-approval year. Premia Spine’s TOPS motion-preservation implant demonstrated superior functional outcomes versus fusion in a 24-month randomized study, pressuring traditional rigid construct. Imaging vendors such as Siemens Healthineers partner with implant companies to co-develop AI-enabled planning suites, further blurring hardware-software borders. Supply-chain resilience also shapes competition, with firms localizing manufacture to avoid tariff exposure and MDR bottlenecks.
Price pressure intensifies as hospitals deploy gain-sharing contracts that reward lower implant costs and high patient-reported outcome scores. Vendors respond by unbundling navigation from implant purchase or offering subscription models that spread capital expense. Simultaneously, surgeons’ rising data literacy encourages evidence-based procurement, favoring firms that furnish real-world performance dashboards. Competitive differentiation is thus transitioning from raw implant design to holistic workflow optimization, clinical proof, and economic transparency across the spinal fusion devices market.
Global Spinal Fusion Devices Industry Leaders
-
Globus Medical
-
Johnson & Johnson Services, Inc.
-
Medtronic plc
-
Stryker Corporation
-
Zimmer Biomet Holdings
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- April 2025: Stryker completed the divestiture of its spinal implants arm to Viscogliosi Brothers, creating VB Spine while retaining Mako Spine robotics
- February 2025: Globus Medical acquired Nevro Corp for USD 250 million, integrating HFX spinal cord stimulation into its product mix.
- October 2024: Vy Spine secured FDA clearance for its 3-D-printed lumbar cage, expanding additive options for personalized surgery.
- September 2024: Medtronic and Siemens Healthineers formed a strategic alliance on imaging-guided spine platforms.
Research Methodology Framework and Report Scope
Market Definitions and Key Coverage
Our study defines the spinal fusion devices market as the global sales value of implants, plates, screws, rods, cages, and bone-graft substitutes specifically designed to permanently join two or more vertebrae to treat instability, deformity, or degenerative disease. The valuation tracks factory-built systems delivered for cervical, thoracic, and lumbar fusion procedures across all care settings, expressed in USD.
Scope exclusions include devices used only for vertebral compression augmentation, electrical stimulation, or motion preservation, which have been left outside this sizing.
Segmentation Overview
- By Product Type
- Cervical Fusion Devices
- Thoracic Fusion Devices
- Lumbar Fusion Devices
- Interbody Cages
- Pedicle Screw Systems
- Bone Graft Substitutes and Others
- By Type of Surgery
- Open Spine Surgery
- Minimally Invasive Spine Surgery
- By Surgical Approach
- Transforaminal Lumbar Interbody Fusion (TLIF)
- Posterolateral Fusion (PLF)
- Other Approaches
- By End-User
- Hospitals
- Ambulatory Surgery Centers (ASCs)
- Specialty & Orthopedic Clinics
- By Geography
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific
- China
- India
- Japan
- Australia
- South Korea
- Rest of Asia-Pacific
- Middle East and Africa
- GCC
- South Africa
- Rest of Middle East and Africa
- South America
- Brazil
- Argentina
- Rest of South America
- North America
Detailed Research Methodology and Data Validation
Primary Research
Semi-structured interviews with spine surgeons, OR nurses, hospital supply managers, and regional distributors across North America, Europe, Asia-Pacific, and the Gulf helped validate average implant mixes, price dispersion after discounts, and the shift toward outpatient minimally invasive fusion. Follow-up surveys captured utilization intentions that filled remaining data gaps.
Desk Research
We began with structured searches of open datasets such as the U.S. FDA 510(k) database, Medicare Outpatient Prospective Payment files, Eurostat hospital discharge records, and trade association registries (e.g., International Society for the Advancement of Spine Surgery). Company 10-Ks, investor decks, and customs shipment logs complemented these statistics, while D&B Hoovers and Dow Jones Factiva supplied financial clues on ASP moves. Select insights from Marklines and Questel patent libraries informed adoption timing of novel expandable cages. The sources listed illustrate, not exhaust, the wider set consulted.
Market-Sizing & Forecasting
A top-down model anchors total procedures using national surgery counts and degenerative spine prevalence ratios, which are then multiplied by weighted device bundles and blended ASPs. Supplier roll-ups and sampled channel checks act as bottom-up reasonableness tests before totals are reconciled. Key variables like aging population growth, elective surgery deferrals, titanium price trends, MIS penetration rates, and regulatory approval velocity drive the model. Multivariate regression with scenario analysis projects 2026-2030 values, guided by consensus ranges gathered during primary research and adjusted where bottom-up evidence diverges beyond 5%.
Data Validation & Update Cycle
Outputs pass a three-layer review: automated variance scans, senior analyst peer checks, and final sign-off. We refresh the model annually; interim events like major reimbursement shifts trigger rapid re-contacts so clients receive the latest view.
Why Mordor's Spinal Fusion Devices Baseline Commands High Trust
Published estimates often differ because analysts select unlike product baskets, price assumptions, and refresh cadences.
Key gap drivers include some studies that lump motion preservation implants with fusion hardware; others apply list rather than negotiated prices or back forecast from vendor press releases without procedure controls; a few refresh every three to five years, missing rapid outpatient migration. Mordor's disciplined scope, dual-path modeling, and yearly updates curb those variances.
Benchmark comparison
| Market Size | Anonymized source | Primary gap driver |
|---|---|---|
| USD 5.58 B (2025) | Mordor Intelligence | - |
| USD 7.32 B (2024) | Trade Journal A | Bundles non-fusion robotics and biologics, limited triangulation |
| USD 9.5 B (2025) | Global Consultancy A | Uses vendor shipment totals at list ASPs, includes navigation capital equipment |
| USD 8.31 B (2025) | Industry Association B | Adjusts for hospital billing codes only, assumes uniform global reimbursement |
These contrasts show that when scope creep, outdated prices, or single-source data enter calculations, valuations inflate. By grounding every step in traceable variables and routine cross-checks, Mordor Intelligence delivers the balanced baseline decision-makers can rely on.


Key Questions Answered in the Report
Which surgical technique is gaining the quickest traction among spine surgeons?
Transforaminal lumbar interbody fusion (TLIF) is being adopted rapidly because it restores disc height through a single posterior corridor while preserving muscle integrity and lowering complication risk.
How are 3-D-printed cages changing spinal fusion practice?
Patient-specific, additively manufactured cages improve endplate contact and promote faster osseointegration, translating to higher fusion reliability and fewer revision procedures.
Why are ambulatory surgery centers viewed as attractive venues for fusion procedures?
ASCs combine shorter turnover times with proven safety records, allowing payers and providers to realize substantial cost savings without sacrificing clinical outcomes.
What impact do bundled payment models have on implant selection?
Hospitals now choose devices based on total episode value, favoring systems that pair strong clinical evidence with lower long-term complication expenses.
How is artificial intelligence influencing intra-operative decision-making?
AI-guided navigation platforms overlay anatomical data in real time, helping surgeons optimize screw trajectories and reduce radiation exposure for both staff and patients.
What is the primary clinical concern limiting unlimited use of rigid fusion constructs?
Long-term studies highlight adjacent-segment disease as a meaningful risk, prompting interest in motion-preserving solutions and hybrid stabilization techniques.
Page last updated on:








