Market Size of South Africa In-Vitro Diagnostics Industry
| Study Period | 2019 - 2029 |
| Base Year For Estimation | 2023 |
| Forecast Data Period | 2024 - 2029 |
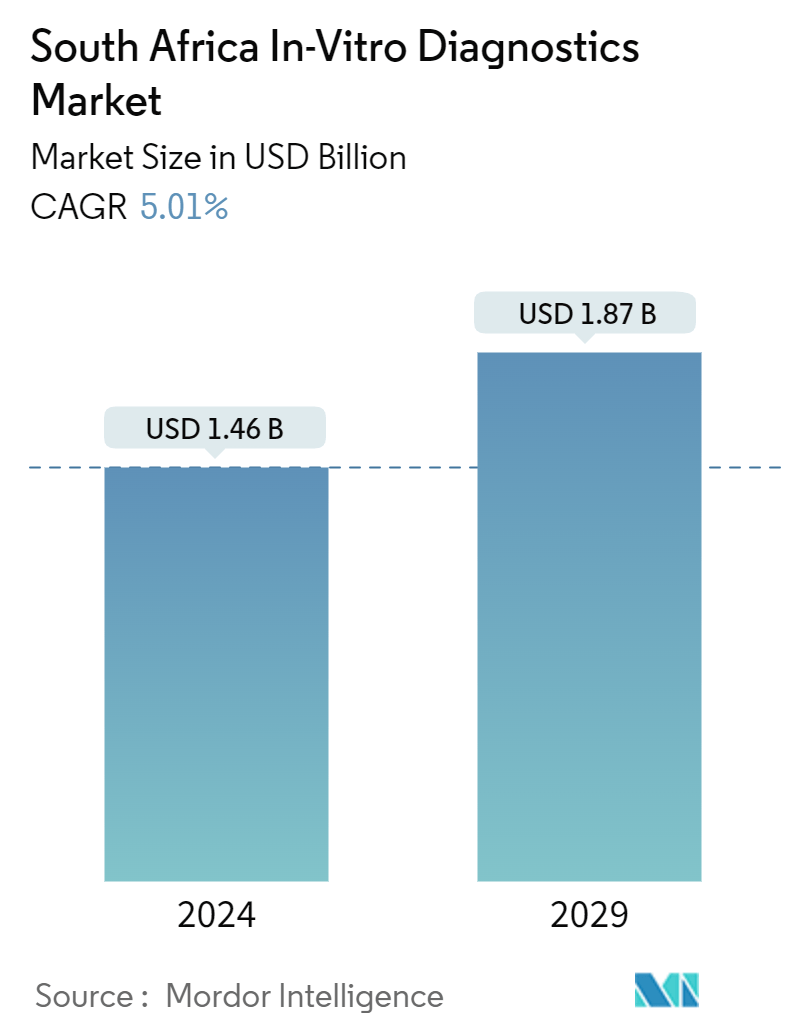
| Market Size (2024) | USD 1.46 Billion |
| Market Size (2029) | USD 1.87 Billion |
| CAGR (2024 - 2029) | 5.01 % |
Major Players
*Disclaimer: Major Players sorted in no particular order |
South Africa In-Vitro Diagnostics (IVD) Market Analysis
The South Africa In-Vitro Diagnostics Market size is estimated at USD 1.46 billion in 2024, and is expected to reach USD 1.87 billion by 2029, at a CAGR of 5.01% during the forecast period (2024-2029).
The COVID-19 pandemic turned the spotlight on the IVD market since there was an increasing demand for IVD kits and reagents for the rapid and accurate diagnosis of SARS-CoV-2 virus infection among the global population. The outbreak of COVID-19 positively impacted the diagnostics industry, as in-vitro diagnostics involved the testing of various biological samples. This aided in the diagnosis of infectious diseases, such as COVID-19. Testing remained a crucial step in controlling the COVID-19 pandemic. Also, the rapid launch of diagnostic kits for the diagnosis of COVID-19 by key players drove the studied market during the pandemic. For instance, according to the data by WHO in February 2022, WHO increased the number of laboratories able to detect COVID-19 from two to more than 900 and bolstered genetic sequencing efforts in Africa through several initiatives including setting up the Regional Centre of Excellence for Genomic Surveillance and Bioinformatics in South Africa in collaboration with South African National Bioinformatics Institute (SANBI) at the University of Western Cape. These efforts led to more than 7,500 samples being sequenced every month in Southern Africa, and led to more than 54% increase in sequencing data on the continent, thereby contributing to the growth of the Africa IVD market. Hence, the COVID-19 pandemic posed a favorable impact on the market initially, and as the pandemic subsided the market lost some traction. However, it is expected to have a stable growth during the forecast period of the study.
The IVD market in South Africa is expected to grow at a good rate over the forecast period. This growth is majorly attributed to the rising burden of chronic and infectious diseases in South Africa, increasing the use of point of care diagnostics in the country, and technological advancements in the IVD industry. According to an article published by PubMed Central in July 2022, the national prevalence of HIV in South Africans was 14.0% across all ages, and it is estimated that around 7.9 million people are affected with the virus. As HIV-affected people are more prone to various chronic and infectious diseases, the demand for in-vitro diagnostics products, including diagnostic testing, is expected to be high in the country for diagnosis and monitoring purposes. In addition, according to the International Diabetes Federation (IDF) 2021 report, the estimated number of South African adults aged 20-79 years with diabetes is expected to increase from 4.2 million in 2021 to 7.5 million by 2045. Therefore, due to the high and increasing burden of diseases, the IVD market is expected to grow over the forecast period with the increase in demand for in-vitro tests and diagnostics products in South Africa.
Additionally, the current market is also growing due to the use of advanced technologies in the IVD market. There has been a paradigm shift from traditional diagnostics to a new generation of diagnostics that work at the gene level. This was made possible by the inclusion of advanced technologies, such as genetic testing, molecular diagnostics, polymerase chain reaction (PCR), and next-generation sequencing (NGS) in the IVD platform.
Also, the increasing product launches with advanced features are expected to drive the studied market. In January 2021, local biotech startup BioCODE, headquartered in Cape Town, started developing a cost-effective test to identify early-stage cancer and cardiovascular disease. In addition, the rapid test will determine an individual's risk for the development of the above-mentioned diseases. By the end of 2022, this test is anticipated to be implemented throughout the nation.
Thus, above mentioned factors are expected to drive the market during study period. However the reimbursement issues and the high cost associated with advanced IVD devices are expected to restrain the growth of the in-vitro diagnostics market in South Africa.
South Africa In-Vitro Diagnostics (IVD) Industry Segmentation
As per the scope of this report, in-vitro diagnostics involves medical devices and consumables that are utilized to perform in-vitro tests on various biological samples. They are used for the diagnosis of various medical conditions, such as diabetes and cancer. The South Africa In-Vitro Diagnostics Market is segmented by Test Type (Clinical Chemistry, Molecular Diagnostics, Immuno Diagnostics, Hematology, and Other Test Types), Product (Instruments, Reagents, and Other Products), Usability (Disposable IVD Devices and Reusable IVD Devices), Application (Infectious Disease, Diabetes, Cancer/Oncology, Cardiology, Autoimmune Diseases, Nephrology, and Other Applications), and End User (Diagnostic Laboratories, Hospitals and Clinics, and Other End Users). The report offers the value (in USD million) for the above segments.
| By Test Type | |
| Clinical Chemistry | |
| Molecular Diagnostics | |
| Immuno Diagnostics | |
| Haematology | |
| Other Test Types |
| By Product | |
| Instrument | |
| Reagent | |
| Other Products |
| By Usability | |
| Disposable IVD Device | |
| Reusable IVD Device |
| By Application | |
| Infectious Disease | |
| Diabetes | |
| Cancer/Oncology | |
| Cardiology | |
| Autoimmune Disease | |
| Nephrology | |
| Other Applications |
| By End-User | |
| Diagnostic Laboratories | |
| Hospitals and Clinics | |
| Other End-users |
South Africa In-Vitro Diagnostics Market Size Summary
The South Africa In-Vitro Diagnostics market is poised for significant growth over the forecast period, driven by the increasing prevalence of chronic and infectious diseases, alongside technological advancements in diagnostic methods. The market experienced a positive impact during the COVID-19 pandemic due to heightened demand for diagnostic kits and reagents, which underscored the critical role of in-vitro diagnostics in disease detection and management. The pandemic also spurred rapid advancements in diagnostic technologies, such as genetic testing and molecular diagnostics, which have since become integral to the market's evolution. Despite the initial boost from COVID-19, the market is expected to maintain stable growth, supported by the rising burden of diseases like HIV and diabetes, which necessitate ongoing diagnostic testing and monitoring.
The market landscape is characterized by a fragmented structure, with numerous global and local players investing in the region to capitalize on growth opportunities. The introduction of advanced diagnostic technologies and the increasing adoption of molecular diagnostic tests are key factors propelling market expansion. Additionally, the reagents segment is anticipated to hold a substantial market share, driven by the demand for chemical, biological, and immunological components essential for in-vitro diagnostics. Regulatory developments and the establishment of a formal medical device regulatory framework in South Africa are expected to further enhance market growth by increasing the usage of diagnostic reagents. As the market continues to evolve, partnerships and collaborations among key players are likely to foster innovation and expand market reach.
South Africa In-Vitro Diagnostics Market Size - Table of Contents
-
1. MARKET DYNAMICS
-
1.1 Market Overview
-
1.2 Market Drivers
-
1.2.1 Increasing Use of Point-of-Care (POC) Diagnostics
-
1.2.2 Growing Burden of Chronic and Infectious Diseases
-
1.2.3 Technological Advancements in the Diagnostics
-
-
1.3 Market Restraints
-
1.3.1 Reimbursement Issues
-
1.3.2 High Cost of Advanced IVD Devices
-
-
1.4 Porter's Five Force Analysis
-
1.4.1 Threat of New Entrants
-
1.4.2 Bargaining Power of Buyers/Consumers
-
1.4.3 Bargaining Power of Suppliers
-
1.4.4 Threat of Substitute Products
-
1.4.5 Intensity of Competitive Rivalry
-
-
-
2. MARKET SEGMENTATION (Market Size by Value - in USD Million)
-
2.1 By Test Type
-
2.1.1 Clinical Chemistry
-
2.1.2 Molecular Diagnostics
-
2.1.3 Immuno Diagnostics
-
2.1.4 Haematology
-
2.1.5 Other Test Types
-
-
2.2 By Product
-
2.2.1 Instrument
-
2.2.2 Reagent
-
2.2.3 Other Products
-
-
2.3 By Usability
-
2.3.1 Disposable IVD Device
-
2.3.2 Reusable IVD Device
-
-
2.4 By Application
-
2.4.1 Infectious Disease
-
2.4.2 Diabetes
-
2.4.3 Cancer/Oncology
-
2.4.4 Cardiology
-
2.4.5 Autoimmune Disease
-
2.4.6 Nephrology
-
2.4.7 Other Applications
-
-
2.5 By End-User
-
2.5.1 Diagnostic Laboratories
-
2.5.2 Hospitals and Clinics
-
2.5.3 Other End-users
-
-
South Africa In-Vitro Diagnostics Market Size FAQs
How big is the South Africa In-Vitro Diagnostics Market?
The South Africa In-Vitro Diagnostics Market size is expected to reach USD 1.46 billion in 2024 and grow at a CAGR of 5.01% to reach USD 1.87 billion by 2029.
What is the current South Africa In-Vitro Diagnostics Market size?
In 2024, the South Africa In-Vitro Diagnostics Market size is expected to reach USD 1.46 billion.

