Market Trends of Risk-Based Monitoring Software Industry
The Service Segment is Expected to Hold a Significant Share in the Market During the Forecast Period
Risk-based monitoring (RBM) represents a shift from traditional, resource-intensive on-site monitoring towards a more targeted and data-driven approach in clinical trial management. Service providers offer expertise in implementing RBM strategies, conducting risk assessments, designing monitoring plans, and managing trial operations.
Service providers offer ongoing support in data analytics, performance monitoring, and risk mitigation strategies throughout the clinical trial. For instance, in an article published by Therapeutic Innovation & Regulatory Science in February 2024, risk-based monitoring (RBM) provides a formal approach to enhancing efficiency, speed, and quality in clinical trials. This is accomplished by prioritizing and mitigating essential safety and efficacy data risks. Hence, as clinical trials increasingly adopt RBM strategies to enhance efficiency, speed, and quality, the demand for specialized services to implement these methodologies is driving growth in the service segment of the risk-based monitoring software market during the forecast period.
Additionally, providers specializing in RBM services offer expertise in risk management, data analytics, and regulatory compliance. RBM services ensure clinical trials' highest quality, accuracy, and validity. For instance, Wemedoo AG's 2024 update stated that the service involves remotely overseeing and analyzing trial data and study activities. Hence, the company's RBM services are crucial in driving growth in the service segment of the risk-based monitoring software market. They provide specialized expertise, ensure data integrity, aid regulatory compliance, enhance operational efficiency, and foster client confidence through a focus on quality and reliability in clinical trial management, which is expected to contribute to the market segment's growth during the forecast period.
Hence, the growing adoption of service providers offering ongoing support in data analytics with the help of RBM and companies focusing on new services for clinical trials is expected to continue in the coming years.
North America is Expected to Hold a Significant Market Share During the Forecast Period
The market's growth in the region is vastly attributed to the rising government investment in clinical trial activity, the presence of major key players, and acquisition by manufacturers in the region.
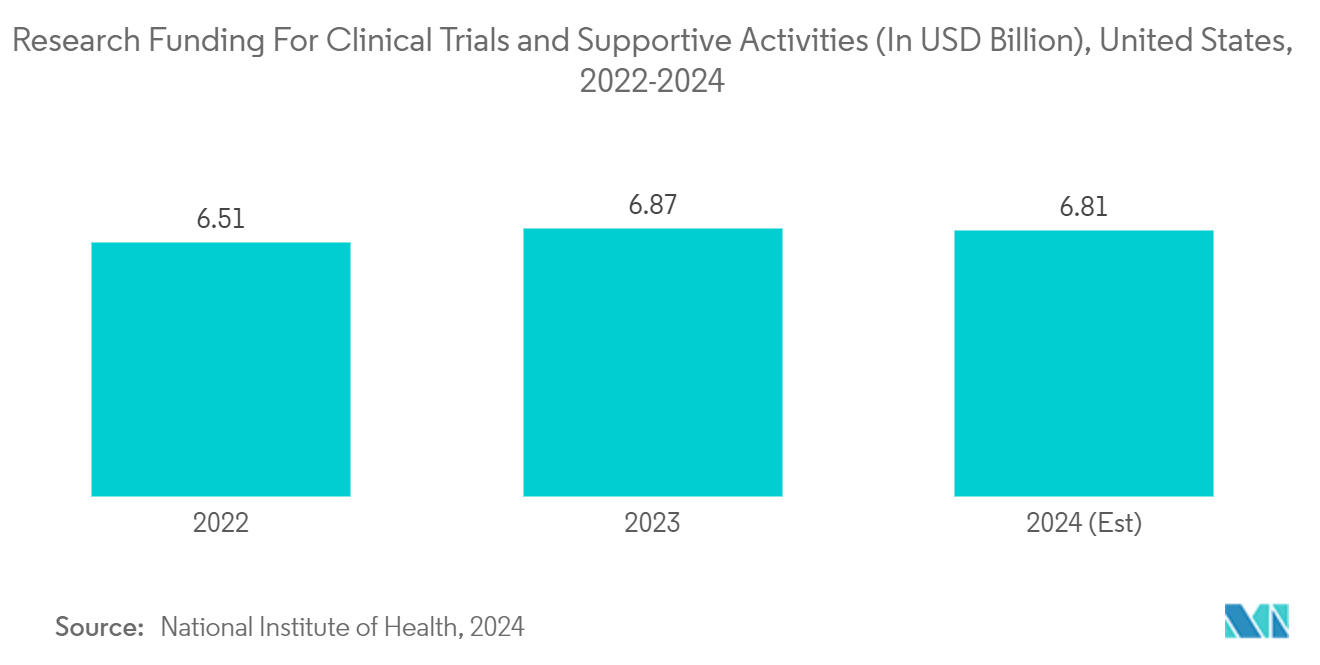
Government investments in clinical trials surge demand for efficient trial management solutions. RBM Software offers advanced analytics and risk-based methodologies that streamline monitoring efforts, enhance data quality, and ensure compliance with regulatory standards. For instance, according to the National Institute of Health, in March 2024, the United States government invested USD 6,878 million in clinical trials and supportive activities in 2023. Thus, government investments in clinical trials promote the adoption of RBM Software and highlight the necessity for robust data management, patient safety, and regulatory compliance, which will further increase the market expansion during the forecast period.
Additionally, strategic partnerships play a crucial role in advancing innovation and efficiency in the evolving landscape of clinical research and pharmaceutical development. For instance, in February 2024, Medidata signed a partnership with pharmaceutical product development (PPD), a clinical research business of Thermo Fisher Scientific Inc. The renewed agreement highlights utilizing the Medidata platform alongside Medidata adjudicate and other rave products. These tools are essential in supporting PPD's efforts to advance drug development programs for its clients. Thus, the partnership between Medidata and PPD strengthens capabilities in clinical trial management and RBM, fostering innovation and efficiency in drug development processes across North America.
Therefore, owing to the aforementioned factors, like government investment in clinical trial activity and manufacturer partnerships, the market is expected to witness significant growth in the region.