Market Trends of Global Point-of-Care Molecular Diagnostics Industry
This section covers the major market trends shaping the Point-of-Care Molecular Diagnostics Market according to our research experts:
The Oncology Segment is Expected to Register a High CAGR During the Forecast Period
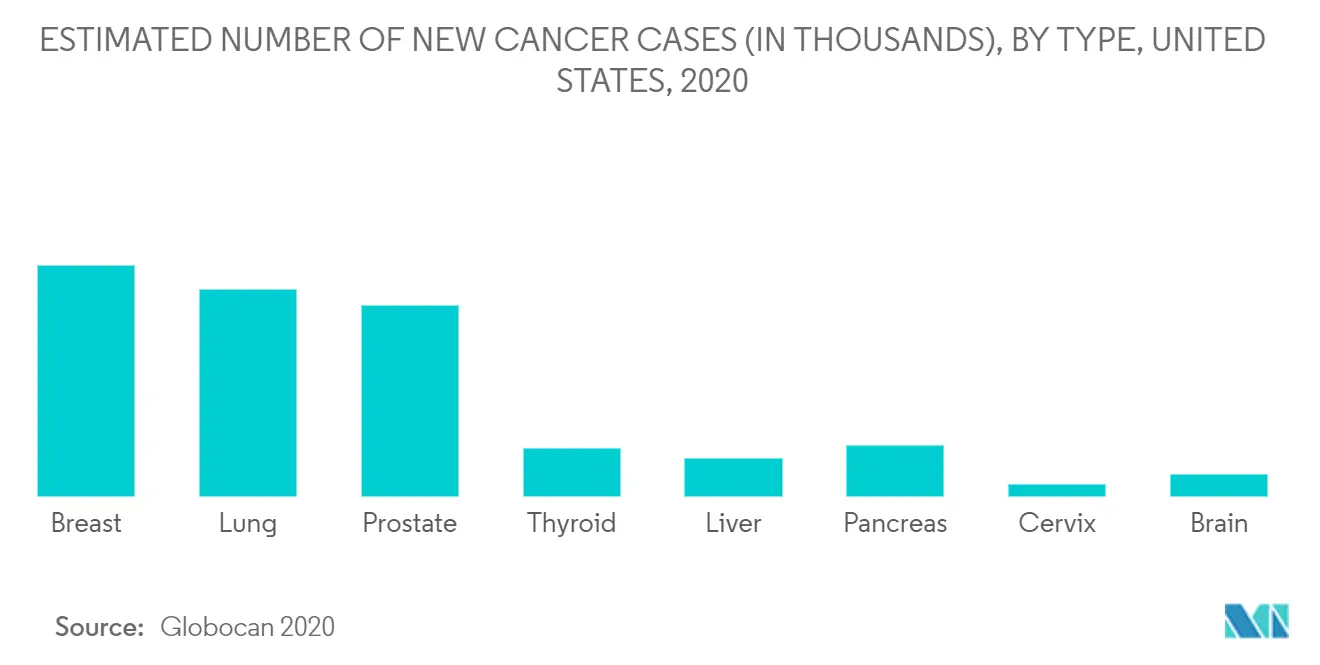
The increasing prevalence of cancer is one of the key reasons for the market's growth. According to the estimates of the Global Cancer Observatory, in 2020, there were 467,965 new cancer cases diagnosed and 185,621 cancer deaths in France. Increasing cancer cases require more diagnoses, thus boosting the Point of Care Molecular Diagnostics Market.
Product launches in the Molecular Diagnostics Industry are driving the market's growth. For instance, in May 2020, a study by the US National Library of Medicine (clinicaltrials.gov) about a trial of one of the Point-of-Care fecal immunochemical testing devices used in the clinical setting was conducted to see if the results are safe and accurate as a ''rule out'' test for colorectal cancer. The study was sponsored by the Royal Surrey County Hospital NHS Foundation Trust. Many current Point of Care Diagnostics utilize lateral flow immunoassay-based technologies. Lateral flow immunoassays are devices that incorporate antibodies to detect the presence of an analyte like cancer biomarkers. In principle, a lateral flow test is a device that will provide a qualitative answer to the presence of a biomarker in a short time, acting as a Rapid Molecular Diagnostic. For instance, in March 2021, Agilent Technologies Inc. entered a definitive agreement to acquire Resolution Bioscience Inc., one of the leaders in the development and commercialization of next-generation sequencing (NGS)-based precision oncology solutions. The acquisition complements and expands Agilent's capabilities in NGS-based cancer diagnostics and provides the company with innovative technology to serve the needs of the fast-growing Molecular Diagnostics Market. Hence, the demand for these quick detection tests, such as Molecular Point of Care Testing, is expected to increase as the incidence of cancer increases.
North America is Expected to Dominate the Market
The prevalence of cancer is the highest in the United States. According to the American Cancer Society, in 2021, there were an estimated 1,898,160 new cancer cases diagnosed and 608,570 cancer deaths in the United States. According to the Global Cancer Observatory, in 2020, there were an estimated 274,364 new cancer cases diagnosed and 86,684 cancer deaths in Canada. The same source also reported that, in 2020, there were an estimated 195,499 new cancer cases diagnosed and 90,222 cancer deaths in Mexico. The prevalence of infectious diseases is also high in the United States and Mexico, creating a high demand for Point of Care Diagnostics tests.
However, the United States has a developed and well-structured healthcare system. The system also encourages R&D. These policies encourage global players to enter the United States, contributing positively to the North America Point of Care Diagnostics Market Outlook. There has also been a considerable influx of product launches that has boosted the Point of Care Molecular Diagnostics Market's growth. For instance, in April 2020, Siemens received FDA clearance for the RapidPoint 500e blood gas analyzer. Such product launch strategies help the company strengthen its position in the POCT Market in the US. The North American region enjoys the presence of many global market players. As high demand is met by the presence of global players in the region, the US POCT Market is further expected to increase.