Market Trends of Plasma Protease C1-inhibitor Industry
This section covers the major market trends shaping the Plasma Protease C1-Inhibitor Market according to our research experts:
Selective Bradykinin B2 Receptor Antagonist Segment is Expected to Witness Healthy CAGR Over the Forecast Period
A bradykinin B2 receptor antagonist used to treat acute episodes of swelling and inflammation associated with hereditary angioedema (HAE). In 2011, Icatibant (Firazyr), a selective bradykinin B2 receptor antagonist, was approved by the US Food and Drug Administration for treatment of acute attacks HAE in adults. Approval was based on 3 double-blind, randomized, controlled clinical trials known as For Angioedema Subcutaneous Treatment (FAST) 1, 2, and 3.
In September 2019, Pharvaris also received a USD 66 Million Series B Financing to Advance the Clinical Development of novel oral B2-receptor antagonists for the treatment of hereditary angioedema (HAE) and other B2-receptor-mediated indications.
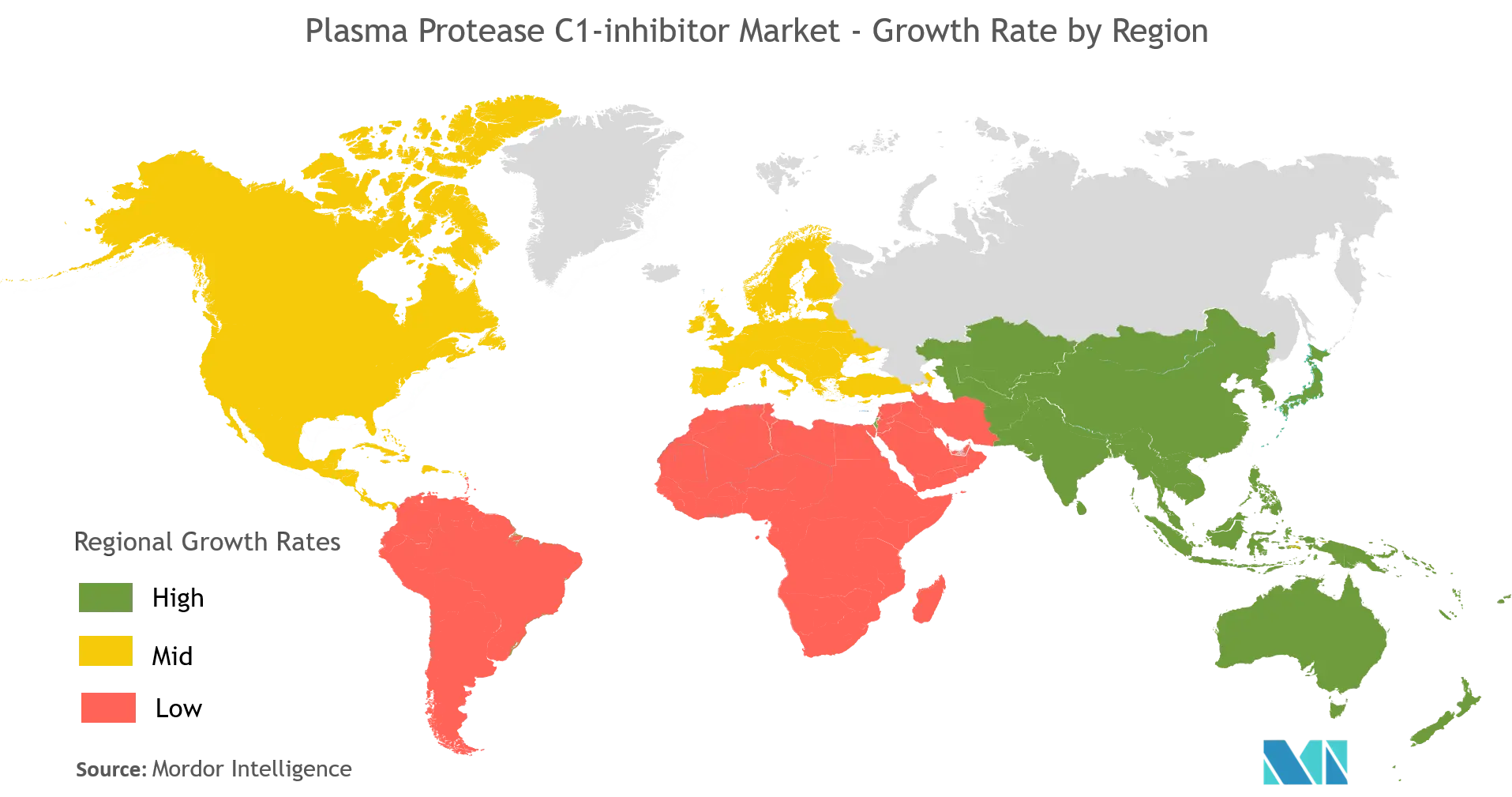
North America is Found Dominating the Global Plasma Protease C1-inhibitor Market
North America dominates the global Plasma Protease C1-inhibitor market owing to the rising Hereditary Angioedema (HAE) and increasing R&D investments. The other factors also includes presence of well-established healthcare infrastructure, an increasing number of product approvals, and major market players providing effective treatment options to the patients suffering from Hereditary Angioedema in North America.
According to DiscoverHAE, it is estimated that 6,000 people in the United States live with HAE currently. In June 2017, CSL Behring received U.S. Food and Drug Administration (FDA) approval for its HAEGARDA (C1 Esterase Inhibitor Subcutaneous [Human]), the first and only subcutaneous therapy indicated for routine prophylaxis to prevent hereditary angioedema (HAE) attacks in adolescent and adult patients. Hence all these factors are expected to drive the overall market in the United States.