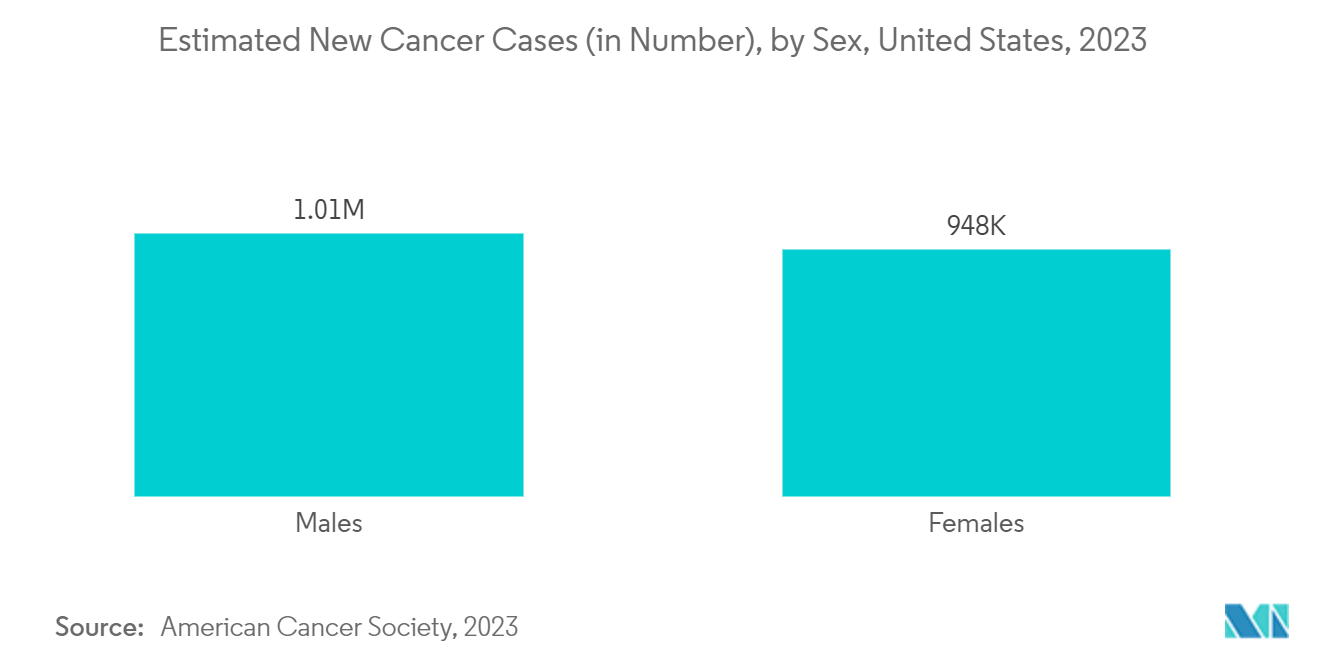Market Trends of North America Companion Diagnostics Devices Industry
The In-situ Hybridization (ISH) Segment is Expected to Exhibit the Fastest Growth Rate over the Forecast Period
The breast cancer segment is anticipated to witness significant growth in the companion diagnostics devices market over the forecast period owing to the factors such as the rising burden of breast cancer cases, growing awareness regarding breast cancer, and increasing new product approvals.
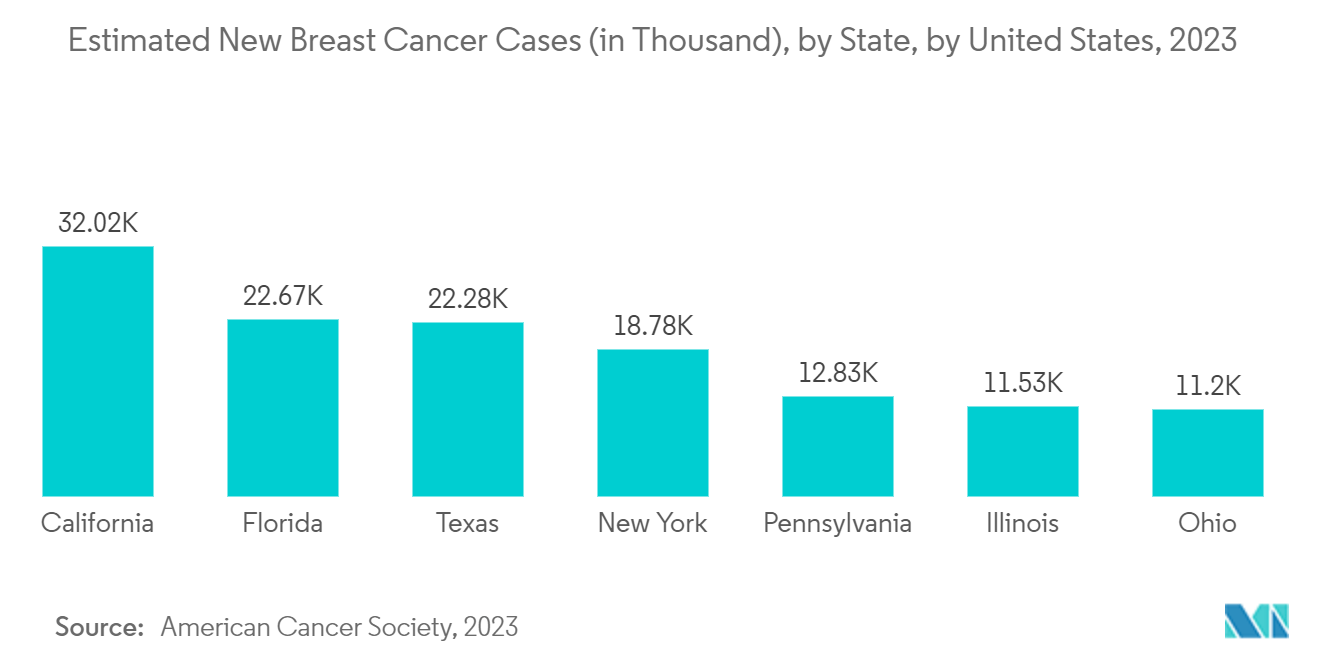
The increasing incidence and prevalence of breast cancer among the population raises the demand for effective and safe administration of a drug or a biological product to treat breast cancer which is only possible by companion diagnostics devices hence propelling the market growth. For instance, according to the statistics published by the Canadian Cancer Society, in May 2022, breast cancer is the most prevalent malignancy and is the second-leading cause of cancer-related death in Canadian women. As per the same source, 28,600 Canadian women are expected to be diagnosed with breast cancer in 2022, which represents 25% of all new cancer cases in women in 2022. Additionally, as per 2023 statistics published by the ACS, about 297,790 new cases of invasive breast cancer and 55,720 new cases of ductal carcinoma in situ (DCIS) are expected to be diagnosed in the United States in 2023.
Furthermore, the rising new product approvals increase the availability of advanced diagnostic devices in the region which is also expected to boost the segment's growth over the forecast period. For instance, in October 2022, Roche received the United States FDA approval for its PATHWAY anti-HER2/neu (4B5) Rabbit Monoclonal Primary Antibody, first companion diagnostics, to identify metastatic breast cancer patients with low HER2 expression for whom Enhertu (fam-trastuzumab deruxtecan-nxki) may be considered as a targeted treatment.
Therefore, due to the aforementioned factors, such as the high burden of breast cancer and increasing new product launches, the studied market is anticipated to grow over the forecast period.
United States is Expected to Have the Significant Market Share Over the Forecast Period
The United States is expected to hold a significant market share in the companion diagnostics market in the North American region over the forecast period owing to the factors such as the rising burden of cancers, growing demand for personalized medicines and targeted therapy, high healthcare expenditure and growing technological advancements in devices.
The increasing incidences and prevalence of various types of cancers in the country raise the need for early detection of cancer and diagnosis of the condition. This is anticipated to fuel the market growth in the country. For instance, as per 2023 statistics published by ACS, about 1,958,310 new cancer cases are expected to be diagnosed in the United States in 2023. In addition, as per the same source, an estimated 238,340 new lung cancer cases, 153,020 new colon/rectum cancer cases, and 297,790 breast cancer cases are expected to be diagnosed in the United States in 2023.
Furthermore, the growing number of new product launches and product approvals increases the availability of novel diagnostics devices in the country, hence propelling market growth. For instance, in August 2022, the United States FDA granted premarket approval to Thermo Fisher Scientific's Oncomine Dx Target test as a companion diagnostic (CDx) to help identify non-small cell lung cancer (NSCLC) patients whose tumors carry epidermal growth factor receptor (EGFR) Exon20-insertion mutations for potential treatment with RYBREVANT targeted therapy. Also, in October 2021, the United States FDA approved Agilent's Ki-67 IHC MIB-1 pharmDx (Dako Omnis), which aids in identifying patients with early breast cancer (EBC) at high risk of disease recurrence.
Therefore, owing to the aforementioned factors, such as the high burden of cancers and new product launches, the studied market is anticipated to grow over the forecast period in the United States.