Market Trends of mRNA Vaccines and Therapeutics Industry
This section covers the major market trends shaping the mRNorth America Vaccines & Therapeutics Market according to our research experts:
Infectious Disease Segment is Expected to Occupy a Significant Share in the mRNA Vaccines and Therapeutics Market over the Forecast Period
The infectious diseases segment is expected to witness significant growth during the forecast period. The increasing prevalence of Ebola, influenza, human immunodeficiency virus (HIV), and other viral infections, including COVID-19, act as major driving factors for this segment. Earlier in 2020, the outbreak of COVID-19 sparked demand for mRNA vaccines and therapeutics. Several major players are actively involved in the research and development of vaccines to combat the COVID-19 infection. COVID-19 has shown a significant positive impact on the infectious diseases segment of the market studied.
Additionally, the growing burden of infectious diseases is expected to propel the infectious diseases segment's growth over the forecast period. According to the Minority of HIV/AIDS Fund (MHAF) United States Department of Health and Human Services Statistics updated in June 2021, there were approximately 37.6 million people living with HIV worldwide in 2020, of whom 35.9 million were adults and 1.7 million were children (under the age of 15). The market is expanding as a result of the rising prevalence of HIV, and the resulting demand for vaccine development is expected to boost the growth of the market over the forecast period.
In addition, non-COVID-19 mRNA vaccines for HIV, influenza, and other viral infections are currently in development and clinical trials. For instance, in April 2022, the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health (NIH), launched a Phase 1 clinical trial to evaluate three experimental messenger RNA (mRNA)-based HIV vaccines platform. The HVTN 302 study is sponsored by NIAID and conducted by the NIAID-funded HIV Vaccine Trials Network (HVTN) based at the Fred Hutchinson Cancer Research Center in Seattle.
Additionally, in July 2021, Moderna announced that the first participants had been dosed in Phase 1 and 2 clinical trials of its quadrivalent influenza candidate, mRNA-1010. Approximately 180 healthy adults in the United States participated in the study and were fully enrolled in early September 2021. Therefore, these increasing mRNA vaccine and therapeutics developments and clinical trials for several infectious diseases are anticipated to grow over the forecast period.
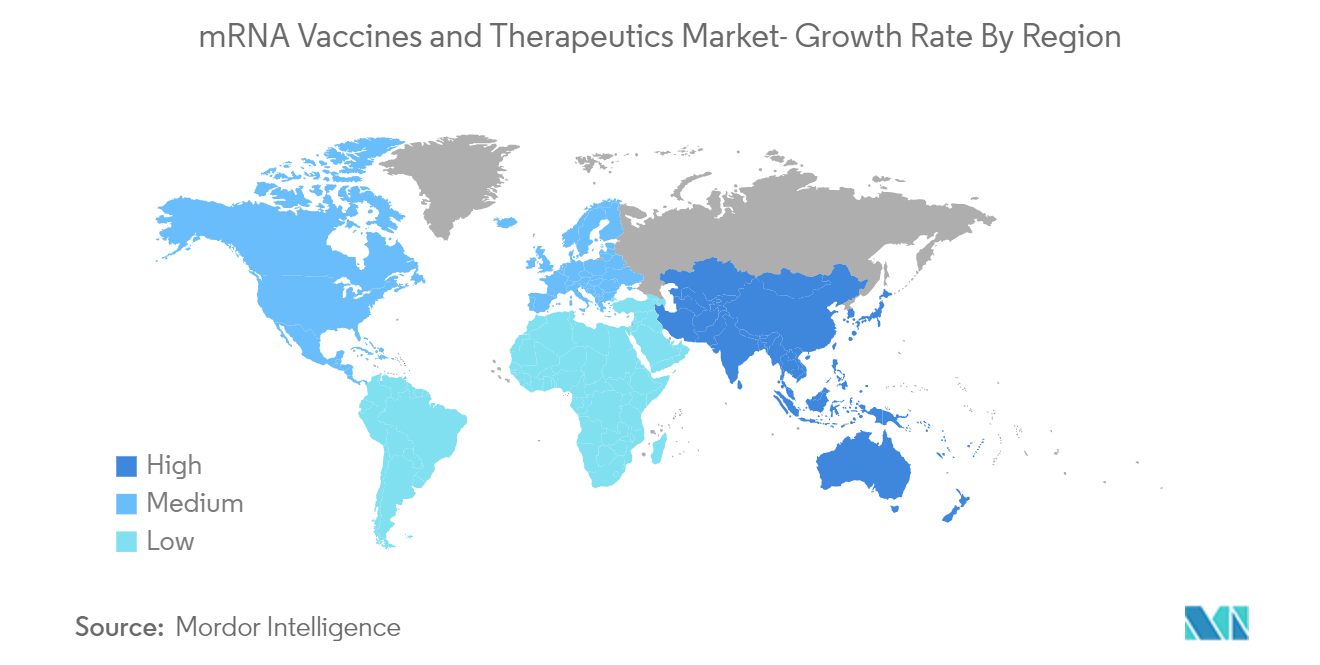
North America is Expected to Hold a Significant Share in the Market in the Forecast Period
North America is expected to dominate the mRNA vaccines and therapeutics market during the forecast period. An increasing number of research and development (R&D) activities and investments in the sector, new product launches, and a rising number of chronic diseases are the major factors attributed to the market's growth in the region. For instance, according to the Cancer Facts & Figures 2022 data published by the American Cancer Society, an estimated 1.9 million new cancer cases were diagnosed and 609,360 cancer deaths in the United States in 2022. Hence, the prevalence of chronic diseases coupled with the increasing geriatric population in the country is anticipated to create more opportunities for the development of new mRNA vaccines in the country, driving the growth of the market studied.
Various initiatives taken by the government are expected to support the market's growth over the forecast period. For instance, in February 2022, the US President's Malaria Initiative invested USD 770 million in 2020 to fight against malaria. In addition, through the Immunization Partnership Fund (IPF), the Government of Canada reached populations with lower vaccine uptake by enabling informed vaccination choices. In 2020 and 2021, IPF received USD 45.5 million in additional funding over 2.5 years to allow the Public Health Agency of Canada to continue supporting vaccination efforts across Canada. Through these initiatives, the Government of Canada is working to increase vaccine confidence, uptake, and access to COVID-19 vaccines across Canada.
Additionally, in May 2021, the Government of Canada announced an investment of up to USD 199.16 million in Resilience Biotechnologies Inc. to increase its manufacturing and filling/finishing capacity for several vaccines and therapeutics that use new technologies such as mRNA. Therefore, health care and drug spending and increasing government activities by these countries are expected to increase in the region over the next few years.
Strategic activities such as new product development, approvals, collaborations, and acquisitions in the United States are driving the growth of the mRNA therapeutics market. For example, in June 2021, Cambridge, Massachusetts, United States-based Strand Therapeutics raised USD 52.0 million to program new cancer therapies. Additionally, as per clinicaltrials.gov, around 150 mRNA vaccine clinical trials were registered in the United States involving biopharmaceuticals in 2022. This increasing number of clinical trials for mRNA vaccines in the country is anticipated to drive the growth of the market studied.