Market Trends of Liver Cancer Therapeutics Industry
Hepatocellular Carcinoma Segment is Expected to Occupy a Significant Share Over the Forecast Period
- Hepatocellular carcinoma (HCC) is a common form of liver cancer that occurs in people suffering from chronic liver diseases like cirrhosis. Since HCC usually grows slowly in its early stages, it can often be cured if discovered early enough with proper treatment options.
- Factors such as an increase in strategic activities by the key players, awareness relating to liver cancer treatments, and a rise in research studies related to the treatments for HCC are expected to bolster segment growth over the forecast period.
- Various therapies have emerged for treating HCC, which made the segment more exposed to research and development. For instance, as per the article published in April 2021 in Nature Journal, the best available first-line treatment for advanced HCC is a combination of PDL1 blockade with atezolizumab and VEGF blockade with bevacizumab. Immunotherapy is likely to synergize with local and locoregional interventions in earlier stages of HCC. Thus, the introduction of new immunotherapies in HCC is expected to bolster segment growth over the forecast period.
- Furthermore, a rise in approvals and strategic activities by the key players is expected to augment the market growth. For instance, in February 2023, Genoscience Pharma received a Food Drug Administration (FDA) Orphan Drug Designation for ezurpimtrostat to treat hepatocellular carcinoma (HCC). Ezurpimtrostat (GNS561) is a first-in-class, first-in-human autophagy inhibitor whose anticancer activity is linked to PPT-1 inhibition.
- Also, in October 2022, the National Medical Products Administration (NMPA) of China approved the supplemental New Drug Application (sNDA) for CYRAMZA (ramucirumab) by Innovent Biologics, Inc., in patients with hepatocellular carcinoma (HCC, also known as liver cancer), who have an alpha-fetoprotein of more than or equal to 400 ng/mL and have been treated with sorafenib. Also, in March 2022, Innovent and Lilly expanded their strategic partnership in oncology.
- Therefore, due to the increase in research activities and strategic initiatives by the key players coupled with product launches, the studied segment is expected to witness significant growth over the forecast period.
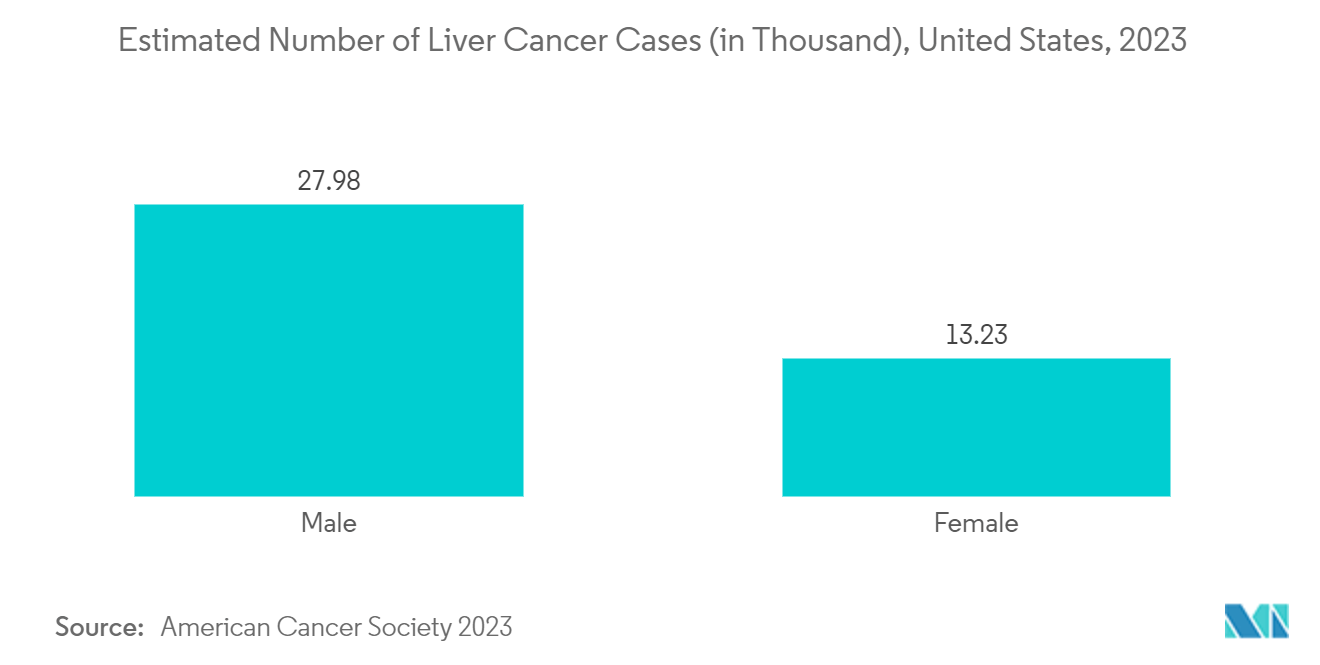
North America is Anticipated to Hold a Significant Market Share Over the Forecast Period
- North America is expected to hold a significant share of the studied market owing to the rising incidence of liver cancer in the region, the presence of key market players, and the launch of novel products coupled with various investments as well as funding for liver cancer therapeutics development. For instance, as per the Canadian Cancer Society 2022 statistics, 3,500 Canadians were diagnosed with liver cancer in Canada.
- The substantial prevalence of liver cancer in the region is anticipated to be the key driving force for the market. As awareness of liver cancer continues to rise and its burden increases, there is a corresponding surge in the adoption and demand for treatment procedures. This is expected to exert a favorable influence on the market under examination.
- For instance, according to the 2023 report of the American Cancer Society, about 41,210 new cases of liver and intrahepatic bile duct cancer are expected to be diagnosed in the United States in 2023, which is expected to have a positive impact on liver cancer therapeutics market. Further, the increasing research and development activities and strategic activities by the key players in the development of novel therapeutics for the liver are also expected to drive market growth.
- For instance, in March 2021, Boston Scientific Corporation received FDA approval for the TheraSphere Y-90 Glass Microspheres, developed for the treatment of patients with hepatocellular carcinoma (HCC). Similarly, in January 2022, Merck received conditional liver cancer treatment approval for its drug Keytruda based on its second confirmatory study. Keytruda slashed the risk of death by 21% over placebo in hepatocellular carcinoma patients in Asia.
- Therefore, the increased incidence of liver cancer and the rise in strategic activities by the key players increase the demand for liver cancer treatment in the studied region, which is driving the market over the forecast period.