Market Size of Global In Silico Clinical Trials Industry
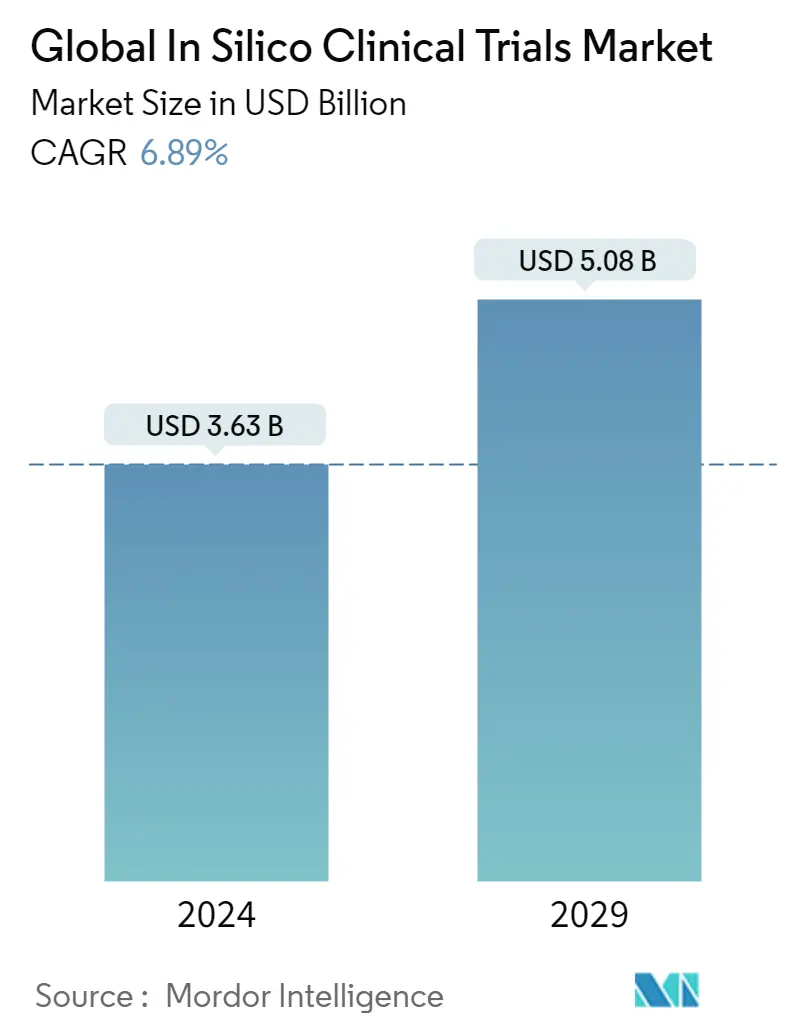
| Study Period | 2019 - 2029 |
| Market Size (2024) | USD 3.63 Billion |
| Market Size (2029) | USD 5.08 Billion |
| CAGR (2024 - 2029) | 6.89 % |
| Fastest Growing Market | Asia-Pacific |
| Largest Market | North America |
| Market Concentration | Low |
Major Players
*Disclaimer: Major Players sorted in no particular order |
In Silico Clinical Trials Market Analysis
The Global In Silico Clinical Trials Market size is estimated at USD 3.63 billion in 2024, and is expected to reach USD 5.08 billion by 2029, growing at a CAGR of 6.89% during the forecast period (2024-2029).
The unanticipated spread of COVID-19 significantly influences the in silico clinical trials market. Even though the news article "Why 2020 Saw The Steady Rise Of In Silico Trials", published in January 2021, explains that as COVID-19-related constraints continue to cause chaos in the clinical research environment, big pharma companies are increasingly turning to in silico or virtual clinical trials to help companies continue their product development efforts. Before evaluating their drug prospects in humans, companies can use advanced computational modeling and simulation tools to test them in "virtual patients." Additionally, since the in silico studies are based on computer simulations, they are unaffected by the ongoing COVID-19-related travel and social distancing restrictions that have hampered many traditional trials.
Furthermore, the proper understanding of drug efficacy and safety, the growing prevalence of numerous diseases, and the cost-effectiveness of in silico clinical trials make the in silico market grow. The article "In silico imaging clinical trials: cheaper, faster, better, safer, and more scalable," published in the Trial journal in 2021, reveals the tremendous advantages offered by in silico clinical trials, such as adjustable variability, unlimited samples, no patient risk, and less burden. These advantages, in turn, lead to high demand for in silico clinical trial market.
Furthermore, market players are engaged in marketing tactics such as mergers and acquisitions and product launches. For instance, in July 2021, Kiromic Biopharma, Inc., a pioneer in Immuno oncology cellular therapy for solid tumors, acquired InSilico Solutions. Kiromic will integrate a team of bioinformatics and AI professionals in-house as part of this deal, extending its lead in the race for AI technology that can identify the best biomarkers for cutting-edge immunotherapeutics like CAR-T cell treatment.
Increased understanding of the benefits of in silico clinical trials has led the market to develop exponentially over the ages. The webcast published by the United States Food and Drug Administration under the title "The VICTRE trial: an in-silico replica of a clinical trial for evaluating digital breast tomosynthesis as a replacement for full-field digital mammography" on March 2021 explains the simulated Virtual Imaging Clinical Trial for Regulatory Evaluation (VICTRE trial) compared digital mammography and digital breast tomosynthesis using computer-simulated imaging of 2,986 in silico patients (an advanced type of mammography that generates three-dimensional images of the breasts using a low-dose x-ray system and computer reconstructions). All breast sizes and lesion types showed an improved lesion detection performance favoring tomosynthesis. The improved tomosynthesis performance was in line with findings from a comparison trial including real patients and radiologists.
As a result of the reasons outlined, the explored market is anticipated to grow throughout the analysis period. However, since human subjects are not involved, the results are based on an approximation that can restrain market growth in silico clinical trials.
In Silico Clinical Trials Industry Segmentation
As per the report's scope, in silico clinical trials refers to the development of patient-specific models to form virtual cohorts for testing the safety and/or efficacy of new drugs and new medical devices. The in silico clinical trials market is segmented by therapeutic area (oncology, infectious disease, cardiology, neurology, diabetes, others), industry (medical devices, pharmaceutical), phase (Phase I, Phase II, Phase III, Phase IV), Geography (North America, Europe, Asia-Pacific, South America, Middle-East, and Africa). The market report also covers the estimated market sizes and trends for 17 countries across major global regions. The report offers the value (in USD million) for the above segments.
| By Therapeutic Area | |
| Oncology | |
| Infectious Disease | |
| Cardiology | |
| Neurology | |
| Diabetes | |
| Other Therapeutic Areas |
| By Industry | |
| Medical Devices | |
| Pharmaceutical |
| By Phase | |
| Phase I | |
| Phase II | |
| Phase III | |
| Phase IV |
| Geography | ||||||||
| ||||||||
| ||||||||
| ||||||||
| ||||||||
|
Global In Silico Clinical Trials Market Size Summary
The in silico clinical trials market is experiencing significant growth, driven by the increasing adoption of virtual trials by pharmaceutical companies. This shift is largely due to the constraints imposed by the COVID-19 pandemic, which highlighted the need for alternative methods to continue drug development without the disruptions caused by travel and social distancing restrictions. In silico trials, which utilize advanced computational modeling and simulation tools, offer a cost-effective and efficient way to assess drug efficacy and safety without the risks associated with traditional human trials. The market is further bolstered by the growing prevalence of various diseases and the technological advancements in artificial intelligence, which enhance the accuracy and reliability of these simulations.
North America holds a dominant position in the in silico clinical trials market, attributed to the region's strong understanding of the benefits and reduced side effects of virtual trials compared to human trials. The presence of key manufacturers and the introduction of innovative products, such as GNS Healthcare's in silico patient for prostate cancer, contribute to the market's expansion. The competitive landscape is moderately active, with major players like Novadiscovery, Insilico Medicine, and GNS Healthcare leading the charge. Collaborations and partnerships, such as those between QuantHealth and 4P-Pharma, are also playing a crucial role in advancing the development and application of in silico trials, further propelling market growth.
Global In Silico Clinical Trials Market Size - Table of Contents
-
1. MARKET DYNAMICS
-
1.1 Market Overview
-
1.2 Market Drivers
-
1.2.1 Better Understanding of the Safety and Efficacy of a Drug
-
1.2.2 Cost-effectiveness and Less Chances of Termination of the Trial
-
-
1.3 Market Restraints
-
1.3.1 Unavailability of Complex Testing Centers and Results Based on Approximation
-
-
1.4 Porter's Five Force Analysis
-
1.4.1 Threat of New Entrants
-
1.4.2 Bargaining Power of Buyers/Consumers
-
1.4.3 Bargaining Power of Suppliers
-
1.4.4 Threat of Substitute Products
-
1.4.5 Intensity of Competitive Rivalry
-
-
-
2. MARKET SEGMENTATION (Market Size by Value - USD million)
-
2.1 By Therapeutic Area
-
2.1.1 Oncology
-
2.1.2 Infectious Disease
-
2.1.3 Cardiology
-
2.1.4 Neurology
-
2.1.5 Diabetes
-
2.1.6 Other Therapeutic Areas
-
-
2.2 By Industry
-
2.2.1 Medical Devices
-
2.2.2 Pharmaceutical
-
-
2.3 By Phase
-
2.3.1 Phase I
-
2.3.2 Phase II
-
2.3.3 Phase III
-
2.3.4 Phase IV
-
-
2.4 Geography
-
2.4.1 North America
-
2.4.1.1 United States
-
2.4.1.2 Canada
-
2.4.1.3 Mexico
-
-
2.4.2 Europe
-
2.4.2.1 Germany
-
2.4.2.2 United Kingdom
-
2.4.2.3 France
-
2.4.2.4 Italy
-
2.4.2.5 Spain
-
2.4.2.6 Rest of Europe
-
-
2.4.3 Asia-Pacific
-
2.4.3.1 China
-
2.4.3.2 Japan
-
2.4.3.3 India
-
2.4.3.4 Australia
-
2.4.3.5 South Korea
-
2.4.3.6 Rest of Asia-Pacific
-
-
2.4.4 Middle-East and Africa
-
2.4.4.1 GCC
-
2.4.4.2 South Africa
-
2.4.4.3 Rest of Middle-East and Africa
-
-
2.4.5 South America
-
2.4.5.1 Brazil
-
2.4.5.2 Argentina
-
2.4.5.3 Rest of South America
-
-
-
Global In Silico Clinical Trials Market Size FAQs
How big is the Global In Silico Clinical Trials Market?
The Global In Silico Clinical Trials Market size is expected to reach USD 3.63 billion in 2024 and grow at a CAGR of 6.89% to reach USD 5.08 billion by 2029.
What is the current Global In Silico Clinical Trials Market size?
In 2024, the Global In Silico Clinical Trials Market size is expected to reach USD 3.63 billion.

