Market Trends of Global PTA Balloon Catheter Industry
This section covers the major market trends shaping the PTA Balloon Catheter Market according to our research experts:
The Coronary Artery Disease Segment is Expected to Hold a Major Market Share in the PTA Balloon Catheter Market
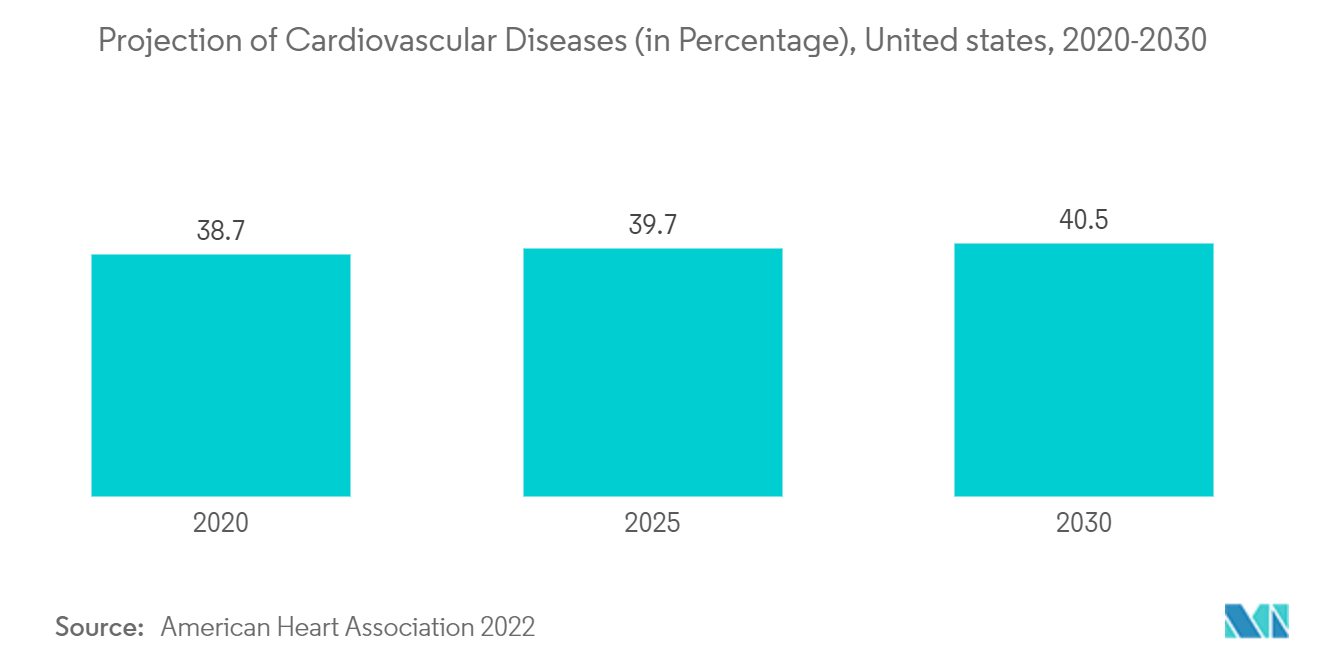
Cardiovascular diseases have become the leading cause of death worldwide in the past three decades. A sedentary lifestyle is one of the major causes leading to many chronic disorders such as diabetes and cardiovascular ailments. Harmful habits such as lack of physical activity, consumption of alcohol and unhealthy food, and smoking have resulted in a growing prevalence of obesity, which in turn leads to various cardiovascular diseases. Surgical intervention for the treatment of such disorders has increased the demand for percutaneous transluminal angioplasty (PTA) balloon catheters. According to a study published by Pub Med Central in 2021, Coronary artery disease is the foremost single cause of mortality and loss of Disability Adjusted Life Years (DALYs) globally. A large number of this burden falls on low and middle-income countries accounting for nearly129 a million DALYs annually, with the growing incidence of coronary artery disease among the people expected to increase demand for catheters, which in turn is expected to drive the growth of the PTA balloon catheter market over the forecast period. For instance, a recent innovation that may offer a paradigm shift in how calcified vessels are treated is the Shockwave Intravascular Lithotripsy system approved by the Food and Drug Administration(FDA) in February 2021 for coronary artery use in February 2021. It combines a low-pressure balloon mounted on a catheter that delivers bursts of sonic energy to crack the calcium.
In addition to this, Boston Scientific Corporation announced it had initiated the AGENT IDE trial for the Agent Drug-Coated Balloon (DCB), the first clinical trial in the United States to evaluate the safety and effectiveness of a DCB in patients with coronary in-stent restenosis (ISR).
Thus, due to the above-mentioned developments, the market is expected to witness significant growth over the forecast period.
North America is Expected to Dominate the Market Over the Forecast Period
The United States is the most affected country across the world, as the increasing prevalence of urological and cardiovascular diseases is expected to increase demand for PTA balloon catheters, which in turn is expected to fuel the PTA balloon catheter market growth over the forecast period.
This can be attributed to factors such as the presence of a large number of PTA balloon catheter manufacturers, suppliers, and distributors, the high prevalence of cardiovascular disorders, the presence of sophisticated healthcare infrastructure, and an increase in patient awareness about emerging minimally invasive surgeries in the region. In addition, the growing preference for minimally invasive surgeries, such as minimally invasive coronary bypass and Minimally Invasive Cardiothoracic Surgery (MICS), is contributing to market growth in North America. The prevalence of cardiac diseases is expected to boost the growth of the market. Also, according to the Centres for Disease Control and Prevention (CDC), in September 2020, heart disease was the leading cardiovascular disease in the United States. Every year about 805,000 Americans has a heart attack. As the number of heart diseases is increasing, there is a growing demand for the proper treatment procedures and devices required to fulfill the unmet needs. Hence are expected to show growth over the forecast period.
Also, the innovative product launches in this region are contributing to the growth of the market. For instance, In June 2021, Neurescue received the 510k clearance from the United States Food and Drug Administration for their NEURESCUE device, 'the world's first intelligent balloon catheter,' devised for the temporary occlusion of large vessels such as the aorta.
Moreover, high expenditure on healthcare, especially in the United States and Canada, along with increased awareness about the disease, is fueling the growth of the overall regional market to a large extent.