Chemotherapy-Induced Neutropenia (CIN) Treatment Market Analysis
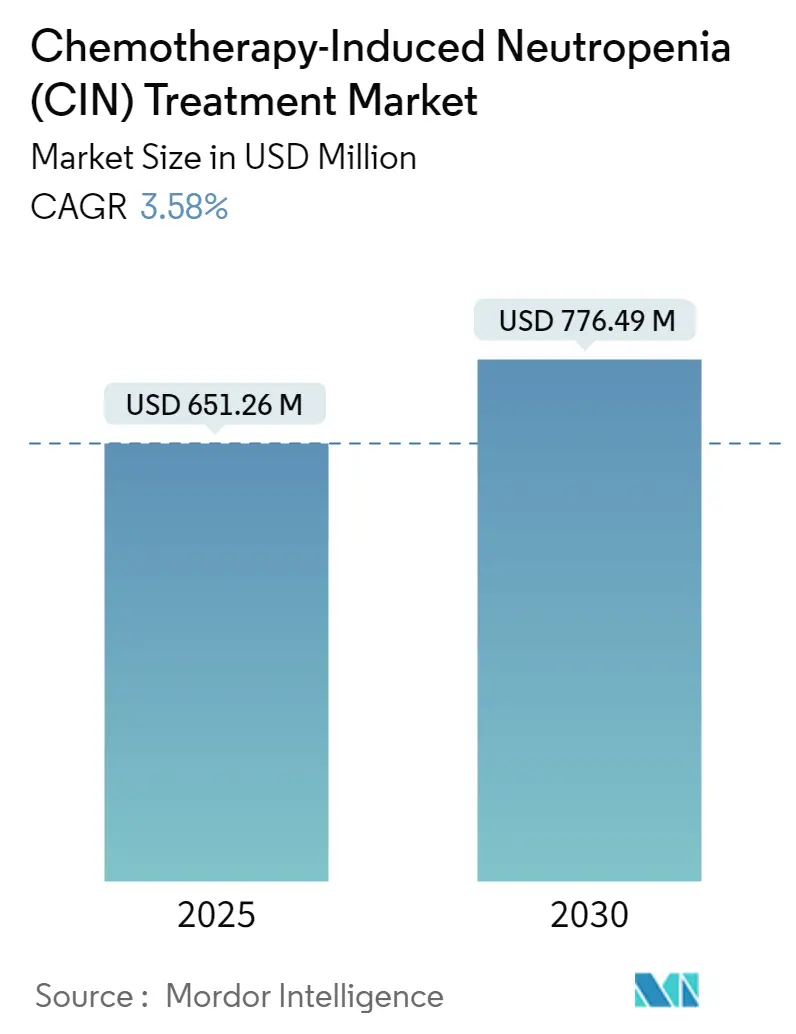
The Chemotherapy-Induced Neutropenia Treatment Market size is estimated at USD 651.26 million in 2025, and is expected to reach USD 776.49 million by 2030, at a CAGR of 3.58% during the forecast period (2025-2030).
The outbreak of COVID-19 impacted the chemotherapy-induced neutropenia (CIN) treatment market. The massive volume of COVID-19 patients and the lockdown disrupted the diagnosis and treatment of non-COVID-19 diseases such as cancer. The decrease in the number of cancers led to a decline in chemotherapy and chemotherapy-induced neutropenia, hampered the demand for chemotherapy-induced neutropenia treatment. For instance, a study published in the European Journal of Epidemiology in January 2023 stated that there had been a decline of over 27.0% in the volume of cancer diagnoses amid the pandemic globally. However, in the post-pandemic period, the volume of COVID-19 patients decreased, which led to the resumption of the diagnosis and treatment of other chronic diseases, such as cancer, and it enabled the market to grow normally. For instance, in February 2021, Tata Memorial Hospital in India reported that the normal count of cancer patients was resumed at the cancer OPD of the hospital after the pandemic. Thus, the pandemic imposed an adverse impact on the chemotherapy-induced neutropenia (CIN) treatment market initially; however, with the decrease in the number of COVID-19 cases, the resumption of the diagnosis and treatment of cancer and the increasing volume of chemotherapy-induced neutropenia (CIN) treatment, the market is growing at a normal pace. It is expected to continue the same over the forecast period.
The increasing prevalence of cancer, rising use of chemotherapy treatment, and increasing research and development activities are the major drivers for the market.
The rising prevalence of cancer and the increasing use of chemotherapy for its treatment has led to an increased use of chemotherapy-induced neutropenia in cancer patients. For instance, as per the American Cancer Society Colorectal Cancer Statistics in 2023, over 153,020 adults are estimated to be diagnosed with colorectal cancer 2023 in the United States. These numbers include 106,970 new cases of colon cancer and 46,050 new cases of rectal cancer in the United States. Furthermore, the data published by the Australian Institute of Health and Welfare in August 2022 stated that over 151,000 new cancer cases were estimated to be diagnosed in Australia in 2021. Thus, such a rising burden of cancer is expected to elevate the number of chemotherapy-induced neutropenia in cancer patients. Therefore, these factors will boost the treatment demand, driving the market growth over the forecast period.
Furthermore, many research studies have highlighted the high risk of developing neutropenia in chemotherapy patients. For instance, an article published in the eCancer Medical Science in February 2021 stated that during chemotherapy, one out of every three patients of breast cancer included in the study developed neutropenia while on chemotherapy. Thus, with the high burden of cancer and the high volume of chemotherapy, the number of chemotherapy-induced neutropenia is expected to increase, which in turn is expected to accelerate the demand for neutropenia treatment products, propelling the market growth.
Moreover, the recent developments by the market players are expected to increase the penetration of treatment products intended to manage chemotherapy-induced neutropenia. For instance, in March 2022, the U.S. FDA approved the Biologics License Application (BLA) of Amneals' filgrastim-ayow, a biosimilar referencing Neupogen used to treat chemotherapy-induced neutropenia. Further, in February 2021, G1 Therapeutics, Inc. received approval from the U.S. FDA for its therapy drug, COSELA (trilaciclib). The drug is intended to manage chemotherapy-induced myelosuppression in adult patients when administered before a platinum/etoposide-containing regimen or topotecan-containing regimen for extensive-stage small cell lung cancer (ES-SCLC).
Therefore, owing to the abovementioned factors, such as the increasing burden of cancer, the high volume of chemotherapy, and the high risk of developing chemotherapy-induced neutropenia, the market is expected to show significant growth over the forecast period.
However, the high cost of neutropenia treatment and strict rules and regulations for product approvals are expected to restrain the growth of chemotherapy-induced neutropenia (CIN) treatment over the forecast period.
Chemotherapy-Induced Neutropenia (CIN) Treatment Market Trends
Granulocyte Colony-Stimulating Factor Therapy Segment is Expected to Witness Significant Growth Over the Forecast Period
Granulocyte colony-stimulating factor therapy (G-CSF) is the most common treatment for cancer-related neutropenia. It is among the most widely prescribed supportive care medications for breast and lymphoma malignancies. The granulocyte colony-stimulating factor therapy segment is anticipated to grow with the advancing research and recent developments by the market players.
Research studies have highlighted the efficacy of the G-CSF therapy, which is anticipated to increase the adoption of the G-CSF therapies for the effective management of cancer-related neutropenia. For instance, a study published in the journal Biology in October 2021 demonstrated that filgrastim and its biosimilars demonstrated to prevent febrile neutropenia in cancer patients. Such studies are expected to increase the demand for G-CSF therapy for treating cancer-related neutropenia, which in turn is expected to boost the segment's growth.
Furthermore, the increasing company focus on developing granulocyte colony-stimulating factor therapy drugs for treating chemotherapy-induced neutropenia in cancer patients through various business strategies such as product launches, agreements, mergers, and collaboration also contribute to the segment growth over the forecast period. For instance, in October 2022, Spectrum Pharmaceuticals received FDA approval for its Rolvedon (eflapegrastim-xnst) to manage non-myeloid malignancies. The drug is a recombinant human granulocyte-colony stimulating factor (GCSF) and is the first novel long-acting GCSF (LA-GCSF) product approved in over 20 years. Furthermore, in February 2022, Lupin signed a license, supply, and technology-sharing agreement with Axantia Holding. Under this agreement, Axantia will be responsible for registering, distributing, and marketing biosimilar Pegfilgrastim drug products in certain territories, including Saudi Arabia, certain GCC countries, Jordan, Lebanon, Iraq, Sudan, Libya, and Algeria.
Therefore, with the increasing adoption of granulocyte colony-stimulating factor therapy (G-CSF) as the primary treatment and increasing company focus on developing effective therapies for treatment against neutropenia in cancer patients, the studied segment is expected to grow over the forecast period.
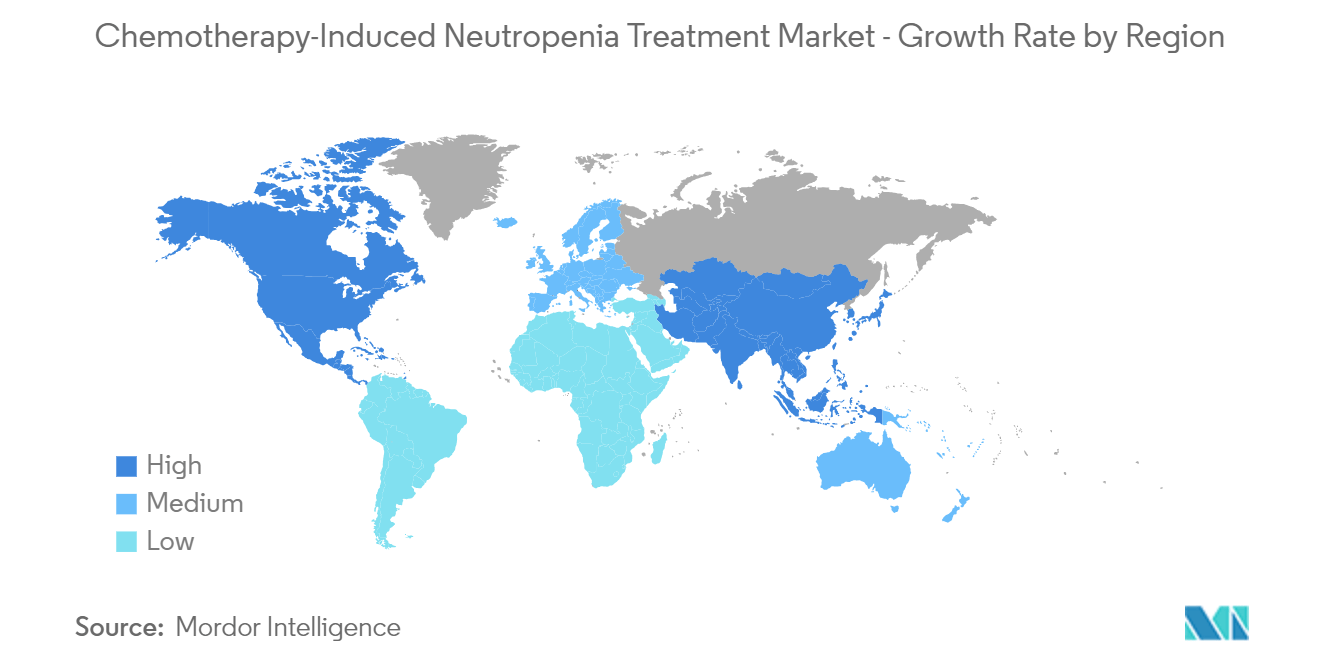
North America is Expected to Witness Significant Growth Over the Forecast Period
North America is anticipated to have a significant market share owing to factors such as the increasing prevalence of cancer, rising demand for disease-specific treatment, high healthcare spending, well-defined infrastructure, and the presence of market players in the region. The rising prevalence of cancer cases in the region is the key driving factor for the market's growth over the forecast period, as chemotherapy is the major cancer treatment, which often further leads to neutropenia. Research studies have highlighted the burden of cancer-related neutropenia in the region, which is expected to boost the demand for chemotherapy-induced neutropenia (CIN) treatment. For instance, according to an article published in the journal Oncologist in August 2022, it is observed that the in-hospital mortality rates for neutropenia/F.N. range from 0.4% to 3.0% for pediatric patients with cancer, 2.6% to 7.0% for adults with solid tumors, and 7.4% for adults with hematologic malignancies. Such instances indicate the need for chemotherapy, which in turn is expected to propel the market growth in the region.
The United States is expected to witness considerable growth among other countries in the North American region. The increasing company focus on developing advanced and efficient therapies for treating chemotherapy-induced neutropenia and increasing drug approvals in the country are the major contributors to the market growth in the country. For instance, in April 2022, Spectrum Pharmaceutical received acceptance for its resubmitted Biologics License Application (BLA) for eflapegrastim by the U.S. FDA for treating chemotherapy-induced neutropenia. Furthermore, in June 2021, BeyondSpring Inc. filed a new drug application, along with a priority review, for its plinabulin in combination with granulocyte colony-stimulating factor (G-CSF) for the prevention of chemotherapy-induced neutropenia (CIN), to the United States Food and Drug Administration.
Thus, the increasing prevalence of cancer, the use of chemotherapy as treatment, and increasing research and development activities in the region are expected to propel market growth over the forecast period.
Chemotherapy-Induced Neutropenia (CIN) Treatment Industry Overview
The chemotherapy-induced neutropenia (CIN) treatment market is consolidated in nature due to the presence of several companies operating globally as well as regionally. The major players operating in the market include Amgen, Inc., Teva Pharmaceuticals Industries Ltd., Merck & Co., Aurobindo Pharma, and BeyondSpring Pharmaceuticals Inc., among others.
Chemotherapy-Induced Neutropenia (CIN) Treatment Market Leaders
-
Amgen, Inc.
-
Teva Pharmaceuticals Industries Ltd.
-
BeyondSpring Pharmaceuticals Inc
-
Aurobindo Pharma
-
Merck & Co.
- *Disclaimer: Major Players sorted in no particular order
Chemotherapy-Induced Neutropenia (CIN) Treatment Market News
- March 2023: Coherus BioSciences received U.S. FDA approval for its pegfilgrastim-cbqv (Udencya), a pegfilgrastim biosimilar for managing febrile neutropenia induced by chemotherapy.
- September 2022: Spectrum Pharmaceuticals, Inc. received U.S. FDA approval for its ROLVEDON (eflapegrastim-xnst) injection, which is intended to decrease the incidence of infection, as manifested by febrile neutropenia in adult patients with non-myeloid-malignancies-receiving myelosuppressive anti-cancer drugs associated with a clinically significant incidence of febrile neutropenia.
Chemotherapy-Induced Neutropenia (CIN) Treatment Industry Segmentation
As per the report's scope, neutropenia is a condition in which the number of white blood cells (neutrophils) in the blood is reduced, lowering the body's ability to fight infections. Chemotherapy-induced neutropenia (CIN) is a common side effect of administering anticancer drugs. This adverse effect has been linked to life-threatening infections and can potentially change the chemotherapy regimen, affecting both short- and long-term outcomes. The Global Chemotherapy-Induced Neutropenia (CIN) Treatment Market is Segmented by Treatment (Antibiotic Therapy, Granulocyte Colony-Stimulating Factor Therapy, Granulocyte Transfusion, and Other Treatments), Distribution channel (Hospital Pharmacies, Retail Pharmacies, and Online Pharmacies), and Geography (North America, Europe, Asia-Pacific, and Rest of the World). The report also covers the estimated market sizes and trends for 17 countries across major regions globally. The report offers the value (in USD) for the above segments.
| By Type | Antibiotic Therapy | ||
| Granulocyte Colony-Stimulating Factor Therapy | |||
| Granulocyte Transfusion | |||
| Other Types | |||
| By Distribution Channel | Hospital Pharmacies | ||
| Retail Pharmacies | |||
| Retail Pharmacies | |||
| Geography | North America | United States | |
| Canada | |||
| Mexico | |||
| Europe | Germany | ||
| United Kingdom | |||
| France | |||
| Italy | |||
| Spain | |||
| Rest of Europe | |||
| Asia-Pacific | China | ||
| Japan | |||
| India | |||
| Australia | |||
| South Korea | |||
| Rest of Asia-Pacific | |||
| Middle East and Africa | GCC | ||
| South Africa | |||
| Rest of Middle East and Africa | |||
| South America | Brazil | ||
| Argentina | |||
| Rest of South America | |||
Chemotherapy-Induced Neutropenia (CIN) Treatment Market Research Faqs
How big is the Chemotherapy-Induced Neutropenia Treatment Market?
The Chemotherapy-Induced Neutropenia Treatment Market size is expected to reach USD 651.26 million in 2025 and grow at a CAGR of 3.58% to reach USD 776.49 million by 2030.
What is the current Chemotherapy-Induced Neutropenia Treatment Market size?
In 2025, the Chemotherapy-Induced Neutropenia Treatment Market size is expected to reach USD 651.26 million.
Who are the key players in Chemotherapy-Induced Neutropenia Treatment Market?
Amgen, Inc., Teva Pharmaceuticals Industries Ltd., BeyondSpring Pharmaceuticals Inc, Aurobindo Pharma and Merck & Co. are the major companies operating in the Chemotherapy-Induced Neutropenia Treatment Market.
Which is the fastest growing region in Chemotherapy-Induced Neutropenia Treatment Market?
Asia-Pacific is estimated to grow at the highest CAGR over the forecast period (2025-2030).
Which region has the biggest share in Chemotherapy-Induced Neutropenia Treatment Market?
In 2025, the North America accounts for the largest market share in Chemotherapy-Induced Neutropenia Treatment Market.
What years does this Chemotherapy-Induced Neutropenia Treatment Market cover, and what was the market size in 2024?
In 2024, the Chemotherapy-Induced Neutropenia Treatment Market size was estimated at USD 627.94 million. The report covers the Chemotherapy-Induced Neutropenia Treatment Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Chemotherapy-Induced Neutropenia Treatment Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Our Best Selling Reports
Chemotherapy-Induced Neutropenia (CIN) Treatment Industry Report
Statistics for the 2025 Chemotherapy-Induced Neutropenia (CIN) Treatment market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Chemotherapy-Induced Neutropenia (CIN) Treatment analysis includes a market forecast outlook for 2025 to 2030 and historical overview. Get a sample of this industry analysis as a free report PDF download.




