Market Trends of Global Biological Safety Testing Industry
This section covers the major market trends shaping the Biological Safety Testing Market according to our research experts:
Bioburden Testing Segment is Expected to Hold Significant Share in the Test Segment of the Market Over the Forecast Period
Bioburden Testing Segment is expected to hold a significant share in the test segment of the market over the forecast period. Organizations like the World Health Organization have also released instructions for health managers and health workers on required infrastructures and standard procedures for effective sterilization, and decontamination of medical devices. These factors are expected to increase the demand for proper sterilization, which is expected to boost the demand for Bioburden testing as it acts as an efficient tool in the validation of validation and revalidation of sterilization processes, assessment of the efficiency of cleaning processes, routine monitoring of manufacturing processes to ensure safety.
Bioburden testing is an integral part of validation and revalidation of sterilization processes, assessment of the efficiency of cleaning processes, routine monitoring of manufacturing processes, monitoring of raw materials, components, or packaging, and overall environmental monitoring programs. The high adoption rate determines bioburden limits in wide-ranging biologics and medical devices. Further, the development of new products in the market is expected to accelerate the market studied. For instance, in December 2019, the company launched the BD Kiestra IdentifA system in Europe and Canada, which is used for microbial identification. It has incorporated automated sample processing steps that ensure accuracy and minimal error.
Furthermore, there are high bioburden and microbial contamination rates during pharmaceutical and biologics manufacturing, which is expected to drive government and private organizations to improve underlying biological safety practices. Hence, all these practices performed might drive the overall market.
North America Dominates the Market and is Expected to do the Same in the Forecast Period
North America Dominates the Market and is expected to do the same in the forecast period. The United States is expected to contribute majorly to the market growth of this region. Due to the outbreak of COVID-19, the rising R&D activities related to COVID-19 have impacted the market growth positively.
The growth of the biological safety testing market in this region can be directly attributed to the growing biotechnology and pharmaceutical industries in the region. The high growth of these industries is further attributable to factors, such as innovative technologies and the presence of favorable government initiatives. Also, the emerging need for validation of drugs and devices process of manufacturing is expected to boost the demand for the market studied. According to the United States Food & Drug Administration (FDA) report, in 2021, the United States had about 4,814 manufacturing sites for drugs.
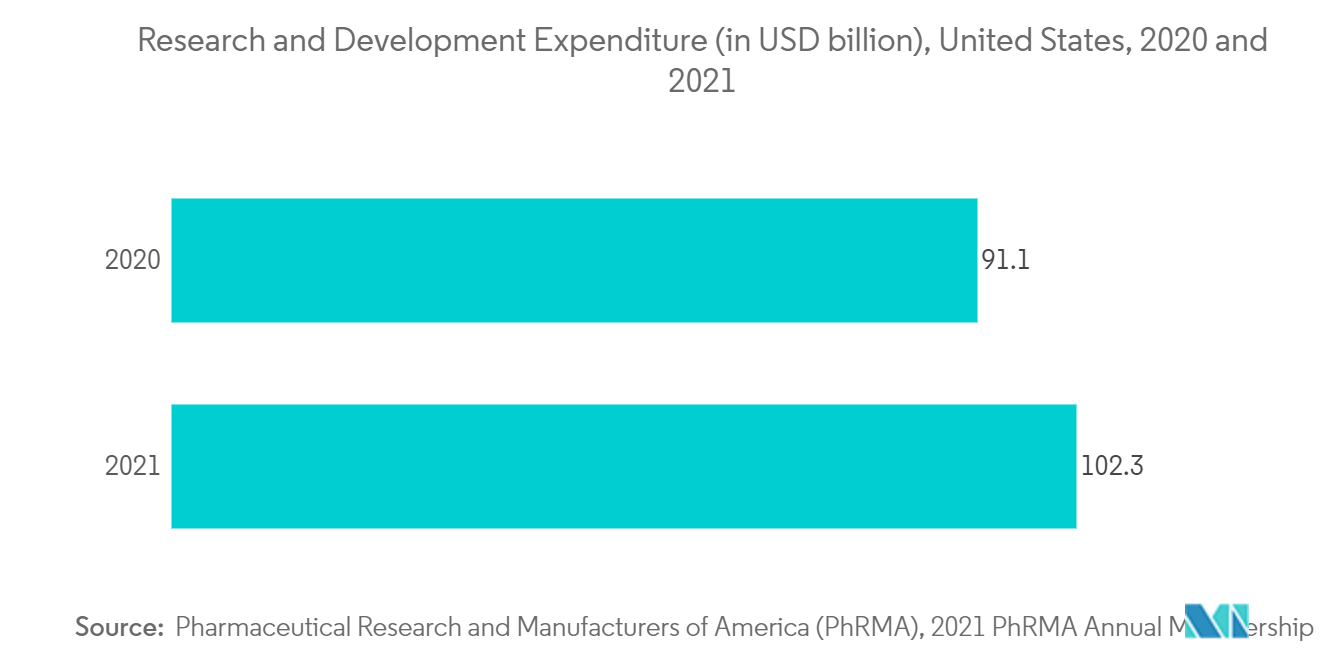
Additionally, the advanced healthcare infrastructure and rising R&D spending are expected to increase the revenue size further. According to the data of Pharmaceutical Research and Manufacturers of America (PhRMA), in the United States, about USD 83 billion were spent by the pharmaceutical industry on the R&D. Thus, this is expected to boost the market growth over the forecast period.