Market Trends of Gene Vector Industry
The Segment for Cancer is Expected to Witness Substantial Growth Over the Forecast Period
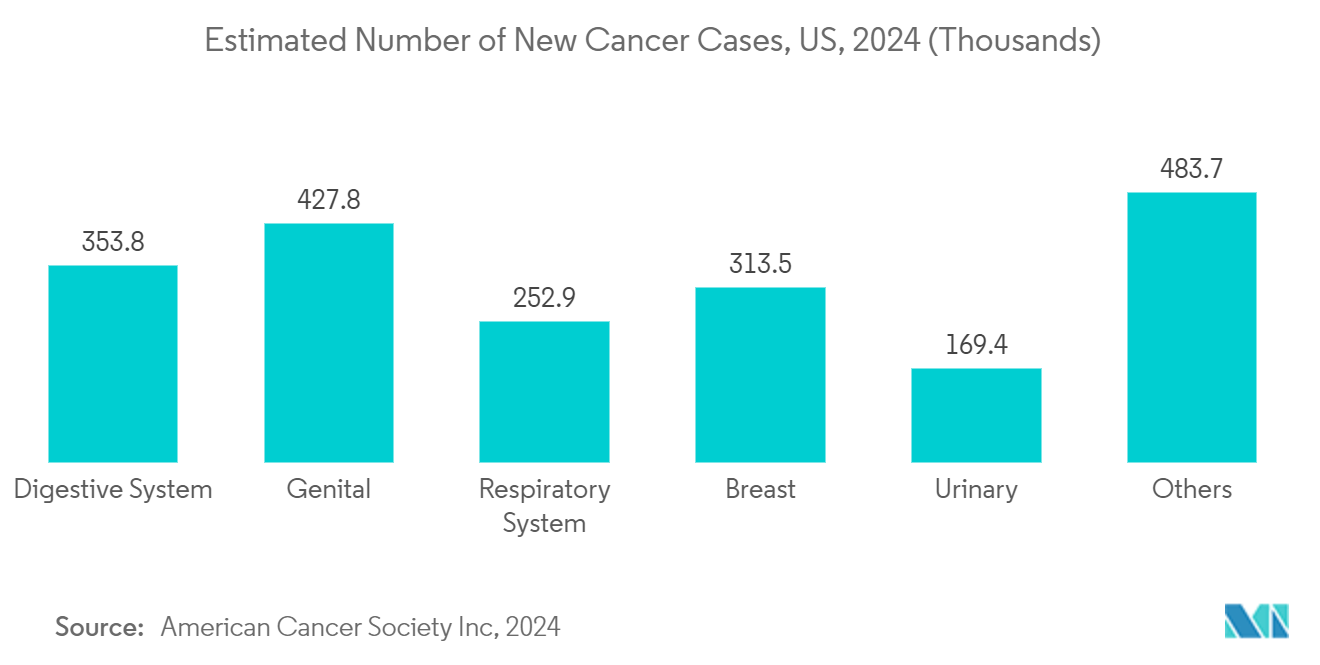
The mounting burden of cancer incidences is a significant health concern witnessed by most countries. The expansion in cancer incidences strongly drives the demand for gene vector-based oncological gene therapy. According to data from June 2022 from the World Health Organization (WHO), every year, approximately 400,000 children develop cancer globally. Similarly, the Indian Journal of Medical Research published a study in March 2023 that estimated the prevalence of cancer in India to rise from 1.46 million in 2022 to 1.57 million by 2025. Owing to such growth in the prevalence of cancer cases, several research programs are ongoing to identify, eliminate, and cure the disease through gene therapy. As of November 2022, the National Clinical Trial (NCT) Registry listed more than 600 ongoing interventional clinical trials related to gene therapy of cancer across different phases of development. Such a growing number of research programs on developing gene therapy drugs promote the growth of this segment.
Varied gene vectors have been engineered in oncology for therapeutic and preventive applications. Several gene therapy strategies are also being developed to treat various cancers, including anti-angiogenesis, oncolytic virotherapy, and therapeutic gene vaccines. For instance, in November 2023, researchers from the University of Texas announced the successful development of a potential gene therapy containing programmed plasmid DNA to "pre-treat" neuroblastoma tumors in order to increase the effectiveness of chemotherapy. Again, in November 2023, new research from the University of Pennsylvania's Gene Therapy Program (GTP) suggested that gene therapy AAVs were unlikely to cause cancer-triggering insertions in humans or monkeys and may contribute to long-term efficacy. Such research findings further promote the application of gene vectors for developing gene therapies to treat cancer. They are also expected to promote the growth of the cancer segment.
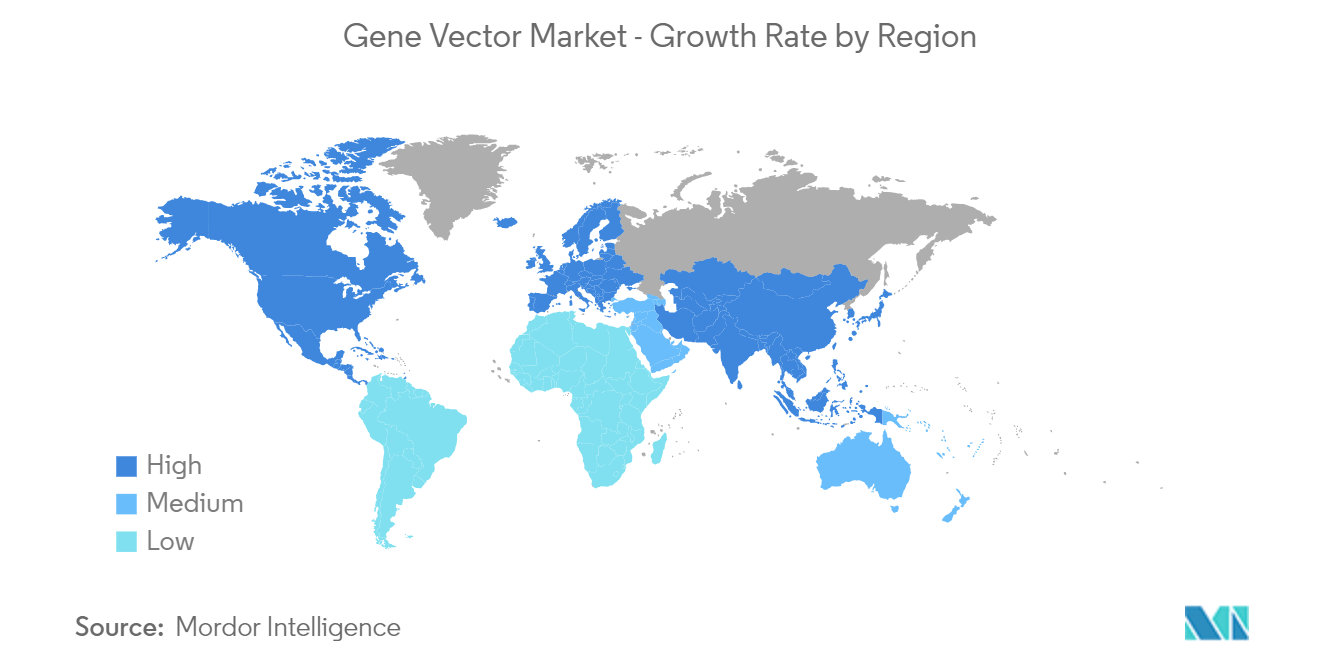
North America is Expected to Dominate the Gene Vector Market
North America is expected to dominate the market owing to factors such as broad regulatory support, competent research institutes, developing healthcare infrastructure, and established players in the region. The United States is renowned for developing medical breakthroughs, owing to government support and funding. The incentives offered through the Orphan Drugs Act have boosted biotechnology and pharmaceutical companies to deliberate advancing research on rare disease medicines as a potentially lucrative venture. According to the Orphan Drug Act, drugs receiving Orphan Drug Designation (ODD) in the country can avail of various regulatory support, including exemption of new drug application fees, tax credits for clinical trial expenses, and 7-year market exclusivity after product approval. Such support for ODD drives the research in various gene therapy drugs, boosting the demand for gene vectors in the United States.
Additionally, there is a constant rise in the number of congenital diseases in the nation. The Institute for Clinical and Economic Review’s (ICER) 2022 report estimated the presence of 7,000 rare diseases in the US, and more than 90% lacked Food and Drug Administration (FDA)-approved disease-specific treatments. It also found that the proportion of new FDA approvals gaining orphan drug designation reached nearly 50% in a bid to curb such a gap. For instance, in December 2023, the FDA granted ODD to NeuExcell Therapeutics Inc.’s NXL-004, an investigational AAV gene therapy product for treating malignant glioma. Also, by 2025, the FDA anticipates approving 10 to 20 cell and gene therapy products yearly. Such factors promote the demand for gene vectors to develop such drugs.
Similar to the US, Canada aims to support healthcare innovation by establishing a pathway for Advanced Therapeutic Products (ATPs). In January 2024, Health Canada approved Pfizer’s BEQVEZ, an AAV vector-based gene therapy for treating adults with moderately severe to severe hemophilia B who are negative for neutralizing antibodies to variant AAV serotype Rh74. In October 2023, Health Canada authorized CSL's HEMGENIX (etranacogene dezaparvovec) as a one-time, single-dose treatment for adults with hemophilia B who require routine prophylaxis. Such approvals indicate the government’s positive outlook toward approving ATPs, which is anticipated to boost the demand for gene vectors in the country.
Therefore, owing to the increasing government support, higher research activities, and rising rate of chronic diseases, the market is expected to witness growth in North America.