Market Trends of Global COVID Rapid Diagnostic Test Industry
This section covers the major market trends shaping the COVID Rapid Diagnostic Test Market according to our research experts:
Molecular Tests Segment is Expected to Register a Significant CAGR Over the Forecast Period
Molecular tests are generally referred to as RT-PCR or nucleic acid amplification tests (NAATs). Bits of viral RNA is amplified by molecular tests to enable the detection of viral infection by specialized testing. Molecular tests primarily include polymerase chain reaction (PCR) tests, loop-mediated isothermal amplification (LAMP), and clustered, regularly interspaced short palindromic repeat (CRISPR)-based assays. Molecular testing offers a higher level of selectivity and is highly precise compared to other tests for COVID-19. Hence, RT-PCR is recommended as the gold standard for COVID-19 testing by several authorities such as WHO, EU, and FDA, among others.
Furthermore, the companies offering COVID-19 detection tests are largely focused on providing highly efficient and precision testing through their innovation, thereby leading to the launch and approval of several molecular diagnostic products, which is expected to positively impact the market. For instance, in May 2022, Belgium-based miDiagnostics launched an ultrafast COVID-19 PCR test based on silicon chip technology. The company's latest test can provide results in about 30 minutes. Also, in March 2022, Sense Biodetection received CE Marking for Veros COVID-19 from the European Authority. Veros COVID-19 is a fully integrated, easy-to-use molecular diagnostic test that provides laboratory-quality results in 15 minutes.
Therefore, due to the high efficacy of molecular tests, and new product launches in the area, the molecular test segment is expected to have a healthy market share.
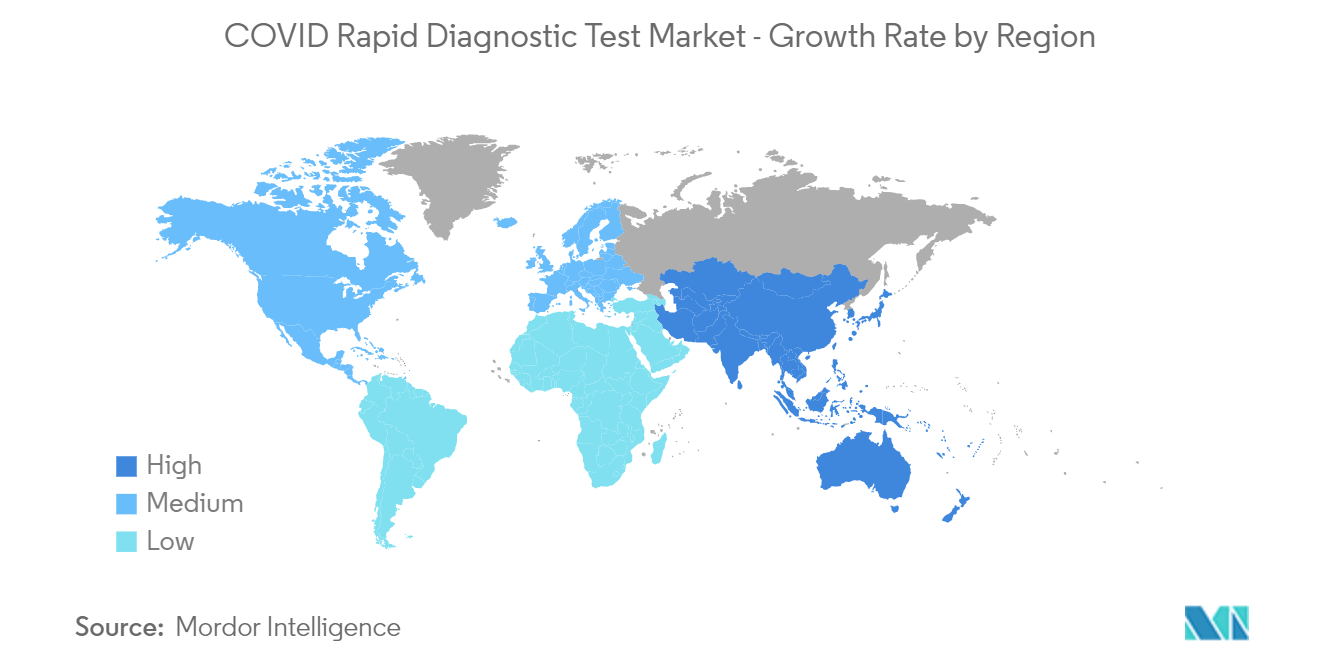
North America is Expected to Hold Significant Market Share Over the Forecast Period
North America is expected to witness significant growth in the COVID rapid diagnostics tests market over the forecast period owing to the factors such as the high prevalence of COVID-19 infections and advanced research and development structure. In addition, the presence of key players as well as rising product launches in the region is also expected to boost the market growth over the forecast period.
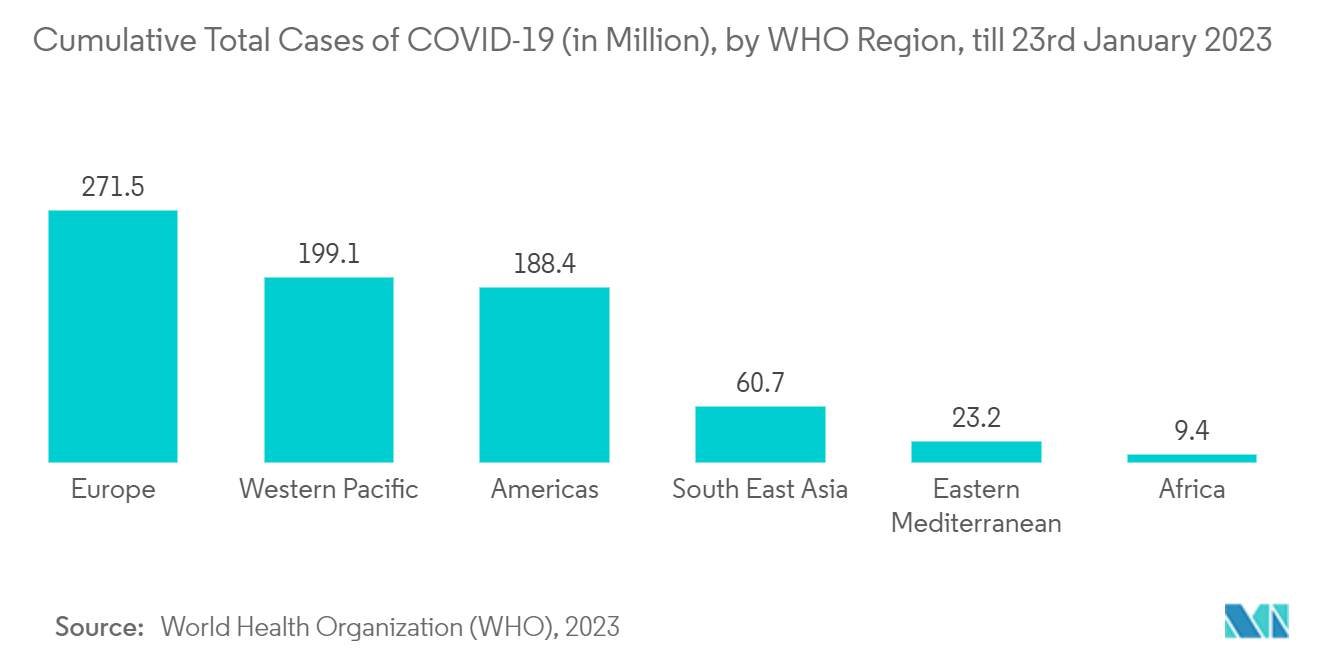
As per WHO COVID-19 Dashboard, as of 18th January 2023, there were about 100.1 million confirmed COVID-19 cases in the United States, which was the highest in the world. This high number of cases creates a demand for COVID-19 detection kits for the rapid detection of the disease, thereby contributing to market growth. Also, as per the same source, about 4,539,229 confirmed cases of COVID-19 in Canada and 7,342,764 confirmed cases of COVID-19 in Mexico were reported from 3rd January 2020 to 27th January 2023. Thus, the high burden of coronavirus infection cases among the population raises the demand for rapid diagnostic tests, thereby propelling market growth.
Additionally, as per an article published in MDPI, in May 2022, it has been observed that, in Mexico, the COVISTIX antigen rapid test is extremely sensitive and specific for detecting Omicron SARS-CoV-2 variant carriers. Also, the test is sufficient for screening both asymptomatic and symptomatic people, including those who have passed the peak of viral shedding and carriers of the extremely common Omicron SARS-CoV-2 subtype. Thus, such a study is anticipated to fuel the demand for rapid antigen test products for detecting emerging variants of COVID-19, hence bolstering market growth.
Moreover, the rising regulatory approvals and new product launches increase the availability of novel diagnostic test kits and devices which in turn is anticipated to propel the market growth over the forecast period. For instance, in December 2022, the Government of Canada approved Btnx's Rapid Response COVID-19 Antigen Self-test Kit. Also, in July 2022, the United States FDA issued an emergency use authorization to Watming's Speedy Swab Rapid COVID-19 Antigen Self-Test, a non-prescription home use for the qualitative detection of nucleocapsid protein antigen from the SARS-CoV-2 virus.
Therefore, owing to the factors, such as the high burden of coronavirus infections and its emerging variants, increasing research studies as well as new product launches, the studied market is anticipated to grow over the forecast period.