Coronary Artery Disease Therapeutics Market Size
| Study Period | 2021 - 2029 |
| Base Year For Estimation | 2023 |
| CAGR | 7.70 % |
| Fastest Growing Market | Asia-Pacific |
| Largest Market | North America |
| Market Concentration | Medium |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Coronary Artery Disease Therapeutics Market Analysis
The Coronary Artery Disease Therapeutics Market is expected to register a CAGR of 7.7% during the forecast period.
The COVID-19 pandemic affected healthcare systems globally and resulted in the interruption of usual care in many healthcare facilities, exposing vulnerable patients with cardiovascular diseases to significant risks. Similarly, the demand for coronary artery disease drugs was significantly impacted during the pandemic owing to the increased risk of COVID-19 infection among patients with cardiovascular diseases. A study published in the Frontiers in Medicine in December 2021 indicated that despite having Coronary artery Disease (CAD) symptoms, 9.1% of patients did not see a medical practitioner due to fear of becoming infected with COVID-19. This led to a decline in market growth initially during the pandemic due to a decrease in the diagnosis of coronary artery disease and the lack of adoption of CAD therapeutics due to stringent lockdowns, supply chain restrictions, and production shutdowns. In the late pandemic, there has been an incline in the market growth of CAD therapeutics due to the rising adoption of CAD therapeutics in COVID-19 patients. For instance, according to the extensive research presented at the American Society of Anesthesiologists annual meeting in October 2022, high cholesterol was treated with statins by 30% of the 38,875 patients hospitalized for COVID-19 at 185 hospitals in the United States between January 1 and September 30, 2020, and found that statin users had a 37 percent lower COVID-19 mortality rate than non-users. In the late pandemic phase, there has been a positive impact on market growth due to the rise in the adoption of statins in COVID-19 patients.
The market is primarily driven by the surge in the prevalence of coronary artery disease across the globe, increasing investment in research and development of coronary artery disease therapeutics, and rising comorbidities such as atherosclerosis and hypertension. The geriatric population is expected to significantly impact the market studied, as people over 65 are more prone to chronic diseases. According to the World Health Organization Facts of 2021, the proportion of the global population aged 60 and up will nearly double from 12% to 22% between 2015 and 2050. By 2050, 80% of the world's elderly will live in low- and middle-income countries. The population is aging at a much faster rate than in the past. According to the British Heart Foundation, in August 2022, Coronary heart disease (CHD) will be the most common type of heart, and circulatory disease in the UK, and 2.3 million people will be living with CHD in 2021. Thus, the high burden of coronary heart disease is expected to increase the demand for its management, thereby contributing to the growth of the market.
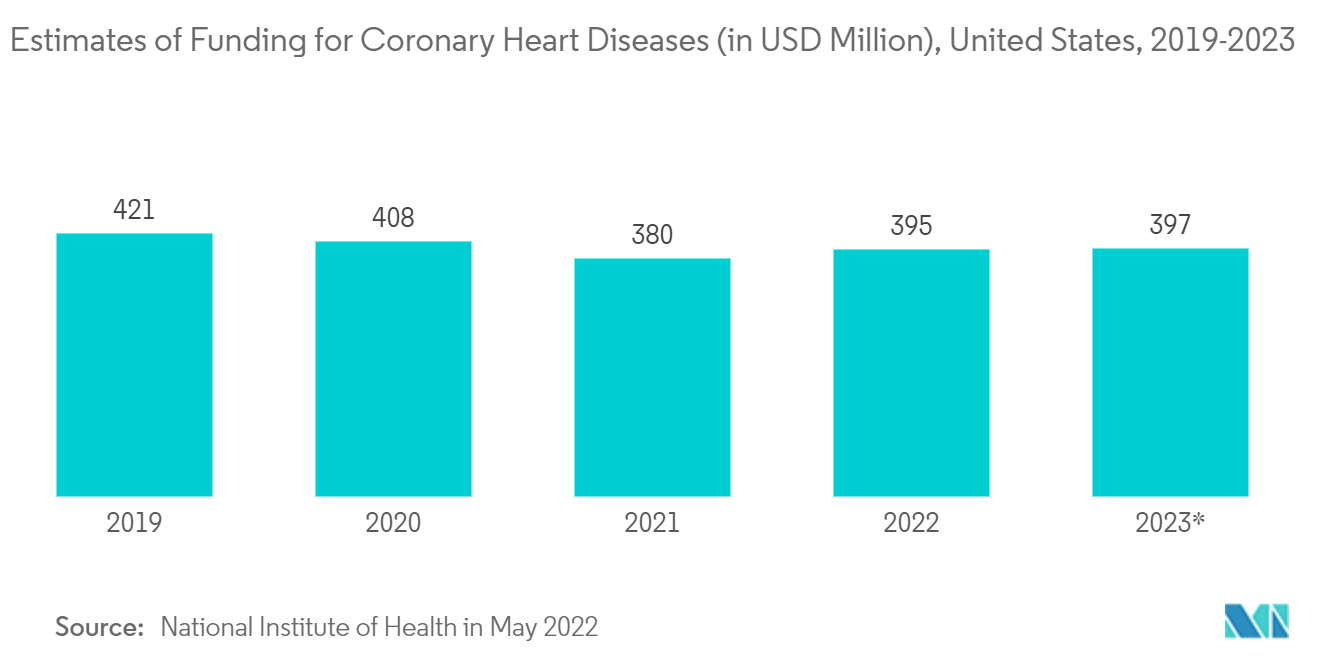
Furthermore, growing incidence and prevalence rates of hypertension, high cholesterol, diabetes, and obesity across the world, government initiatives to increase awareness of the disease, and increasing investment in research and development activity are the key driving factors in the coronary artery disease therapeutics market. For instance, according to the National Institute of Health (NIH) 2022, the estimated NIH funding for Coronary Heart Diseases in 2021 and 2022 is USD 380 million and USD 395 million, respectively. Therefore, the rising funding for CAD will lead to increased research on CAD therapies, thereby leading to innovative therapies' development and market growth.
In addition, rising initiatives from the key market players are also expected to drive market growth over the forecast period. For instance, in December 2021, Novartis entered a commercial agreement with the NHS in England to address elevated LDL-C in eligible patients with coronary artery diseases across England. With this collaboration, the NHS and Novartis move into the implementation phase following the positive final recommendation from the National Institute for Health and Care Excellence (NICE) for the use of inclisiran in primary care for the treatment of adult patients with CAD. Such initiatives are expected to drive market growth due to the rise in the adoption of inclisiran in CAD in developed countries such as the UK.
Therefore, the rising prevalence of CAD, rising initiatives from the market players, the rising geriatric population, and rising research on CAD therapeutics are the factors expected to drive market growth. However, the high cost of treatment, limited awareness, and unavailability of proper healthcare infrastructure for therapeutics may restrain the market growth over the forecast period.
Coronary Artery Disease Therapeutics Market Trends
This section covers the major market trends shaping the Coronary Artery Disease Therapeutics Market according to our research experts:
Statins Segment is Expected to Hold a Significant Market Share in the Coronary Artery Disease Therapeutics Market Over the Forecast Period.
Statin medications are drugs that help to lower cholesterol levels in the blood and prevent coronary heart disease. Statins are the first-line treatment of choice for patients with coronary heart disease and high cholesterol levels. These drugs have additional benefits such as improvements in endothelial function, reduction in the amount of inflammation and damage done to cells through oxidation stress, and prevention of platelet aggregation, thereby reducing the risk of a blood clot (thrombus).
The statins segment holds a significant market share in the coronary artery disease therapeutics market. It is anticipated to show a similar trend over the forecast period due to its ability to reduce mortality and cardiovascular events in patients with coronary artery disease or at high risk of cardiovascular disease. According to the study published in the Journal of Cardiothoracic Imaging in April 2022, baseline statin medication was related to a reduction in major cardiovascular events in people with non-obstructive coronary artery disease (CAD). Statins act by lowering levels of low-density lipoprotein cholesterol, reducing the substrate for plaque formation, and can also stabilize plaques by favorably changing the plaque pathophysiology. Thus, the above benefits of statin therapy in the CAD are expected to increase demand for statins, thereby boosting segment growth.
Moreover, rising approvals of the statins are further expected to boost the market growth. For instance, in March 2021, the FDA approved Althera Pharmaceutical's Roszet (rosuvastatin/ezetimibe) in patients with primary non-familial hyperlipidemia to reduce low-density lipoprotein cholesterol (LDL-C). Such approvals are anticipated to drive segment growth due to the rise in the adoption of statins.
Therefore, the rising approvals of statins and the importance of statins as a first-line treatment in the management of CAD are the factors expected to drive segment growth.
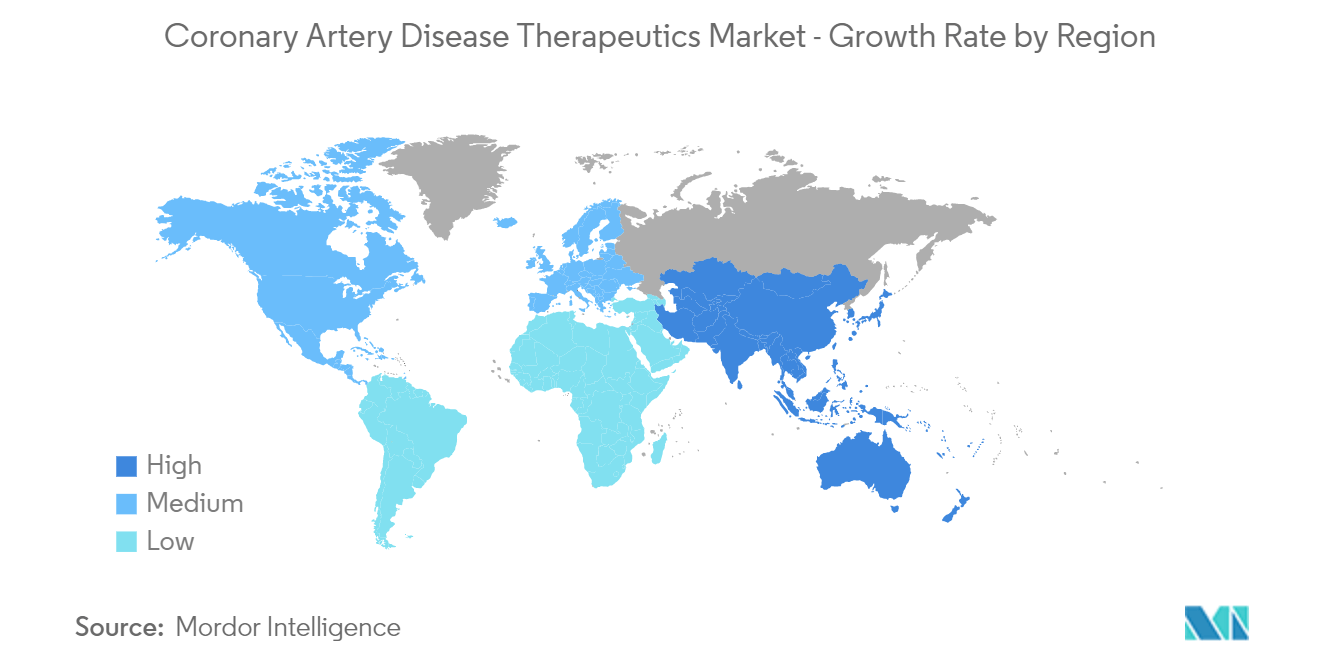
North America is Expected to Hold a Significant Share in the Market and Expected to do Same in the Forecast Period
North America is expected to hold a major market share in the global coronary artery disease therapeutics market due to the growing geriatric population, strong foothold of major market players, and increasing incidences of coronary artery disease.
The rising prevalence of coronary artery disease in the United States is expected to drive the growth of the market studied in the region. For instance, according to CDC, in 2022, the percentage of coronary heart disease for adults aged 18 and over in the United States 2021 is 4.9%. Such a huge prevalence is anticipated to drive market growth due to the rising adoption of therapies in these patients.
Moreover, continuous product approval in the segment is expected to drive segment growth. For instance, in August 2021, Health Canada approved MYINFLA 0.5 mg for the reduction of cardiovascular risk in patients with coronary disease. In addition, in February 2022, FDA approved Norliqva (amlodipine) oral solution for the treatment of hypertension in adults and children 6 years and older and coronary artery disease. Thus, such advances in the region are anticipated to drive market growth over the forecast period.
Thus, the market is expected to witness a high growth rate over the forecast period due to the factors mentioned above in the North American region. Furthermore, high healthcare spending and the presence of well-established healthcare infrastructure are also fueling the growth of the overall regional market to a large extent.
Coronary Artery Disease Therapeutics Industry Overview
The Coronary Artery Disease Therapeutics Market is moderately fragmented and consists of several major players. The competition is driven by the rise in product launches, the rise in partnerships among hospitals and drug developers, and increasing research for the development of novel therapies. Some of the companies which are operating in the market are AstraZeneca Plc, Novartis AG, Pfizer Inc, Bayer AG, Bristol-Myers Squibb Company, Kowa Pharmaceuticals America, Inc., Merck & Co., Inc, Teva Pharmaceutical Industries Ltd, Viatris Inc (Mylan N.V.)
Coronary Artery Disease Therapeutics Market Leaders
AstraZeneca Plc
Novartis AG
Pfizer Inc
Bayer AG
Kowa Pharmaceuticals America, Inc.
*Disclaimer: Major Players sorted in no particular order
Coronary Artery Disease Therapeutics Market News
- May 2022: Zydus Lifesciences introduced bempedoic acid, an oral, non-statin lipid-lowering medication for cardiovascular diseases, under the brand name Bemdac in India.
- May 2022: Amgen released positive topline data from the Phase 2 clinical study of olpasiran in adult patients with Lipoprotein(a) and evidence of atherosclerotic cardiovascular disease (ASCVD).
Coronary Artery Disease Therapeutics Market Report - Table of Contents
1. INTRODUCTION
1.1 Study Assumptions and Market Definition
1.2 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
4.1 Market Overview
4.2 Market Drivers
4.2.1 Surge in Prevalence of Coronary Artery Disease Across the Globe
4.2.2 Increasing Investment on R&D of Coronary Artery Disease Therapeutics
4.2.3 Rising Comorbidities such as Atherosclerosis and Hypertension
4.3 Market Restraints
4.3.1 High Costs of Treatment
4.3.2 Limited Awareness and Unavailability of Proper Healthcare Infrastructure for the Therapeutics
4.4 Porter Five Forces
4.4.1 Threat of New Entrants
4.4.2 Bargaining Power of Buyers/Consumers
4.4.3 Bargaining Power of Suppliers
4.4.4 Threat of Substitute Products
4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value - USD million)
5.1 By Drug Class
5.1.1 Statins
5.1.2 Beta-blockers
5.1.3 Calcium Channel Blockers
5.1.4 ACE Inhibitors
5.1.5 Antiplatelet Drugs
5.1.6 Others
5.2 By Distribution Channel
5.2.1 Hospitals
5.2.2 Online Pharmacies
5.2.3 Retail Pharmacies
5.3 Geography
5.3.1 North America
5.3.1.1 United States
5.3.1.2 Canada
5.3.1.3 Mexico
5.3.2 Europe
5.3.2.1 Germany
5.3.2.2 United Kingdom
5.3.2.3 France
5.3.2.4 Italy
5.3.2.5 Spain
5.3.2.6 Rest of Europe
5.3.3 Asia-Pacific
5.3.3.1 China
5.3.3.2 Japan
5.3.3.3 India
5.3.3.4 Australia
5.3.3.5 South Korea
5.3.3.6 Rest of Asia-Pacific
5.3.4 Middle-East and Africa
5.3.4.1 GCC
5.3.4.2 South Africa
5.3.4.3 Rest of Middle-East and Africa
5.3.5 South America
5.3.5.1 Brazil
5.3.5.2 Argentina
5.3.5.3 Rest of South America
6. COMPETITIVE LANDSCAPE
6.1 Company Profiles
6.1.1 AstraZeneca Plc
6.1.2 Covis Pharma GmbH
6.1.3 Novartis AG
6.1.4 Pfizer Inc
6.1.5 Bayer AG
6.1.6 Bristol-Myers Squibb Company
6.1.7 Kowa Pharmaceuticals America, Inc.
6.1.8 Merck & Co., Inc
6.1.9 Teva Pharmaceutical Industries Ltd
6.1.10 Viatris (Mylan N.V.)
6.1.11 Sanofi
6.1.12 Rosemont Pharmaceuticals
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
Coronary Artery Disease Therapeutics Industry Segmentation
Coronary artery disease is characterized by a narrowing or obstruction of the coronary arteries (blood vessels that carry blood and oxygen to the heart). Atherosclerosis is the most common cause of coronary heart disease (a buildup of fatty material and plaque inside the coronary arteries). The disease is characterized by chest pain, shortness of breath during exertion, and heart attacks. The Coronary Artery Disease Therapeutics Market is segmented by Drug Class (Statins, Beta-blockers, Calcium Channel Blockers, ACE Inhibitors, Antiplatelet Drugs, and Others), Distribution Channel (Hospitals, Online Pharmacies, and Retail Pharmacies), and Geography (North America, Europe, Asia-Pacific, Middle East, and Africa, South America). The market report also covers the estimated market sizes and trends for 17 different countries across major regions globally. The report offers the value (in USD million) for the above segments.
| By Drug Class | |
| Statins | |
| Beta-blockers | |
| Calcium Channel Blockers | |
| ACE Inhibitors | |
| Antiplatelet Drugs | |
| Others |
| By Distribution Channel | |
| Hospitals | |
| Online Pharmacies | |
| Retail Pharmacies |
| Geography | ||||||||
| ||||||||
| ||||||||
| ||||||||
| ||||||||
|
Coronary Artery Disease Therapeutics Market Research FAQs
What is the current Coronary Artery Disease Therapeutics Market size?
The Coronary Artery Disease Therapeutics Market is projected to register a CAGR of 7.70% during the forecast period (2024-2029)
Who are the key players in Coronary Artery Disease Therapeutics Market?
AstraZeneca Plc, Novartis AG, Pfizer Inc, Bayer AG and Kowa Pharmaceuticals America, Inc. are the major companies operating in the Coronary Artery Disease Therapeutics Market.
Which is the fastest growing region in Coronary Artery Disease Therapeutics Market?
Asia-Pacific is estimated to grow at the highest CAGR over the forecast period (2024-2029).
Which region has the biggest share in Coronary Artery Disease Therapeutics Market?
In 2024, the North America accounts for the largest market share in Coronary Artery Disease Therapeutics Market.
What years does this Coronary Artery Disease Therapeutics Market cover?
The report covers the Coronary Artery Disease Therapeutics Market historical market size for years: 2021, 2022 and 2023. The report also forecasts the Coronary Artery Disease Therapeutics Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
Coronary Artery Disease Therapeutics Industry Report
Statistics for the 2024 Coronary Artery Disease Therapeutics market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Coronary Artery Disease Therapeutics analysis includes a market forecast outlook to 2029 and historical overview. Get a sample of this industry analysis as a free report PDF download.
