China In-vitro Diagnostics (IVD) Market Size
| Study Period | 2021 - 2029 |
| Base Year For Estimation | 2023 |
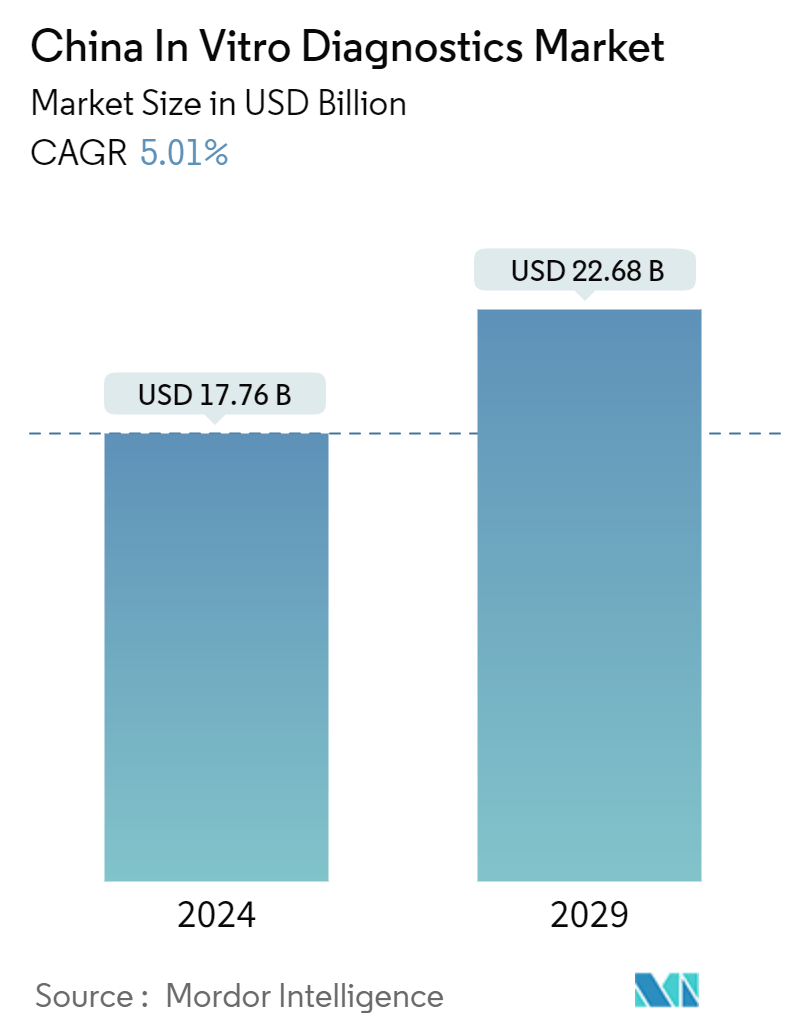
| Market Size (2024) | USD 17.76 Billion |
| Market Size (2029) | USD 22.68 Billion |
| CAGR (2024 - 2029) | 5.01 % |
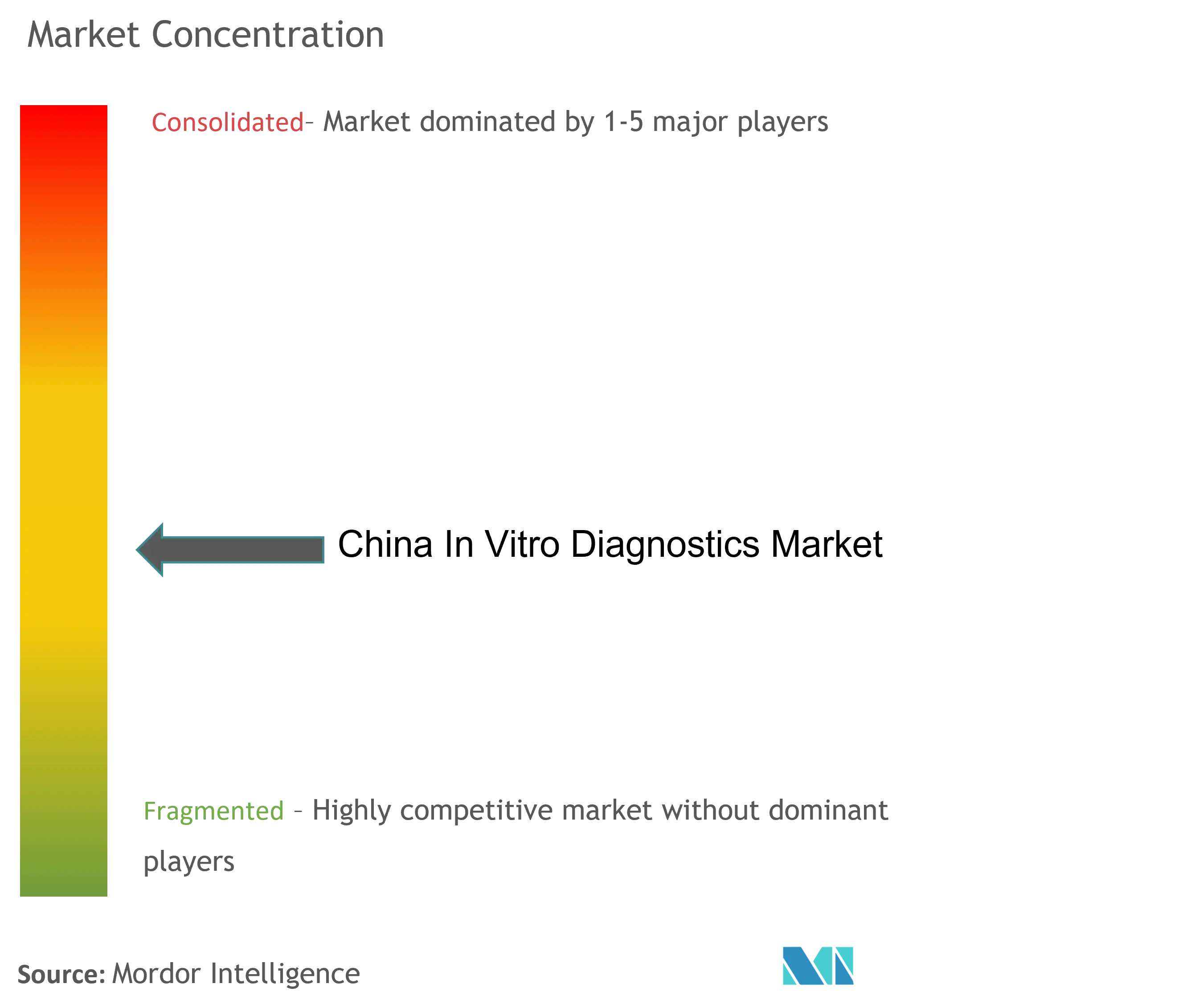
| Market Concentration | Medium |
Major Players
*Disclaimer: Major Players sorted in no particular order |
China In-vitro Diagnostics (IVD) Market Analysis
The China In Vitro Diagnostics Market size is estimated at USD 17.76 billion in 2024, and is expected to reach USD 22.68 billion by 2029, at a CAGR of 5.01% during the forecast period (2024-2029).
The COVID-19 pandemic turned the spotlight on in vitro diagnostics, significantly impacting the COVID-19 in vitro diagnostics market, since there was an increasing demand for IVD kits and reagents for the rapid and accurate diagnosis of SARS-CoV-2 virus infection among the global population. The outbreak of COVID-19 positively impacted the market, as in vitro diagnostics involved the testing of various biological samples. This aided in the diagnosis of infectious diseases such as COVID-19. Testing remained a crucial step in controlling the COVID-19 pandemic. For instance, according to the WHO, 102,762 COVID-19 cases were recorded as of March 2021, with 4,851 deaths in China.
The increasing burden of the COVID-19 pandemic led to a high requirement for tests, which boosted the IVD market in the country. However, the stabilizing cases of COVID-19 in China were expected to stabilize the growth of the market. For instance, as per the National Health Commission (NHC), in December 2022, China reported 36,061 new COVID-19 infections, of which 4,150 were symptomatic and 31,911 were asymptomatic. Thus, the stabilizing cases of COVID-19 were expected to bring the growth of China's in vitro diagnostics market back to its pre-pandemic level.
In vitro diagnostics (IVDs) play an important role in detecting infectious diseases. A large number of molecular assays and immunoassays are being used for the detection of COVID-19. China represents one of the largest China in vitro diagnostics markets in the Asia-Pacific region, and this trend is expected to continue over the forecast period. Due to an increase in the geriatric population and the high burden of chronic and infectious diseases, like diabetes, cancer, and COVID-19, the market studied is slated to show significant growth. This China IVD market growth is expected as these chronic disorders can be diagnosed and monitored using IVD products.
In May 2021, the State Council issued a guideline to implement the country's Healthy China initiative to diminish the incidence of disability among elderly people aged between 65 and 74 years by 2022 by offering medical and health services. Such initiatives by the government are expected to boost market growth during the forecast period. But a lack of proper reimbursement and strict regulations could slow the growth of the market.
Also, the increasing number of product launches with advanced features is expected to drive the market. For instance, in September 2023, Shanghai Kehua Bio-engineering Co., Ltd, a leading China diagnostics manufacturer, has obtained the IVDR CE certificate of Class D high-risk rapid diagnostic product for HIV(1+2) Antibody (Colloidal Gold) V2.
Due to these and other factors, the China IVD market is expected to grow over the next few years.
China In-vitro Diagnostics (IVD) Market Trends
Molecular Diagnostics Segment Dominates the Chinese In Vitro Diagnostics Market
In diagnostics research, one of the most valuable advancements has been in the form of molecular diagnostic tools. The polymerase chain reaction is at the forefront of molecular diagnostics, providing essential diagnostic tests for detecting diseases. The real-time PCR products simultaneously detect viruses, bacteria, fungi, and parasites, allowing molecular laboratories to operate at lower costs and ensure better outcomes, making them essential diagnostic equipment in the fight against infectious diseases.
Molecular diagnostic tests look for specific sequences in DNA or RNA, such as single nucleotide polymorphisms (SNPs), deletions, rearrangements, insertions, and others, that may or may not be linked to a disease, using specialized diagnostic reagents.
Molecular diagnostics were mainly used to diagnose infectious diseases, and with the rising burden and increasing spread of the COVID-19 pandemic, the segment was at its peak in the country. Molecular diagnostic tests, a critical component of the China in vitro diagnostics market, were used to find viral RNAs that are specific to the SARS-CoV-2 virus, which causes COVID-19.
In addition, the rising burden of infectious disease can be attributed to the segment's growth. For instance, according to the National Health Commission, nearly 1.71 million cases of infectious diseases were reported on the mainland. Approximately 288,739 cases were classified as Class B infectious diseases. Viral hepatitis, TB, syphilis, scarlet fever, and gonorrhea accounted for 94% of these cases. Thus, owing to the rising importance given by clinical laboratories to diagnose diseases in the early stages of development, collaboration with domestic manufacturers, and new product launches, the molecular diagnostics segment is expected to witness good growth over the forecast period in China, contributing to the China IVD market growth.
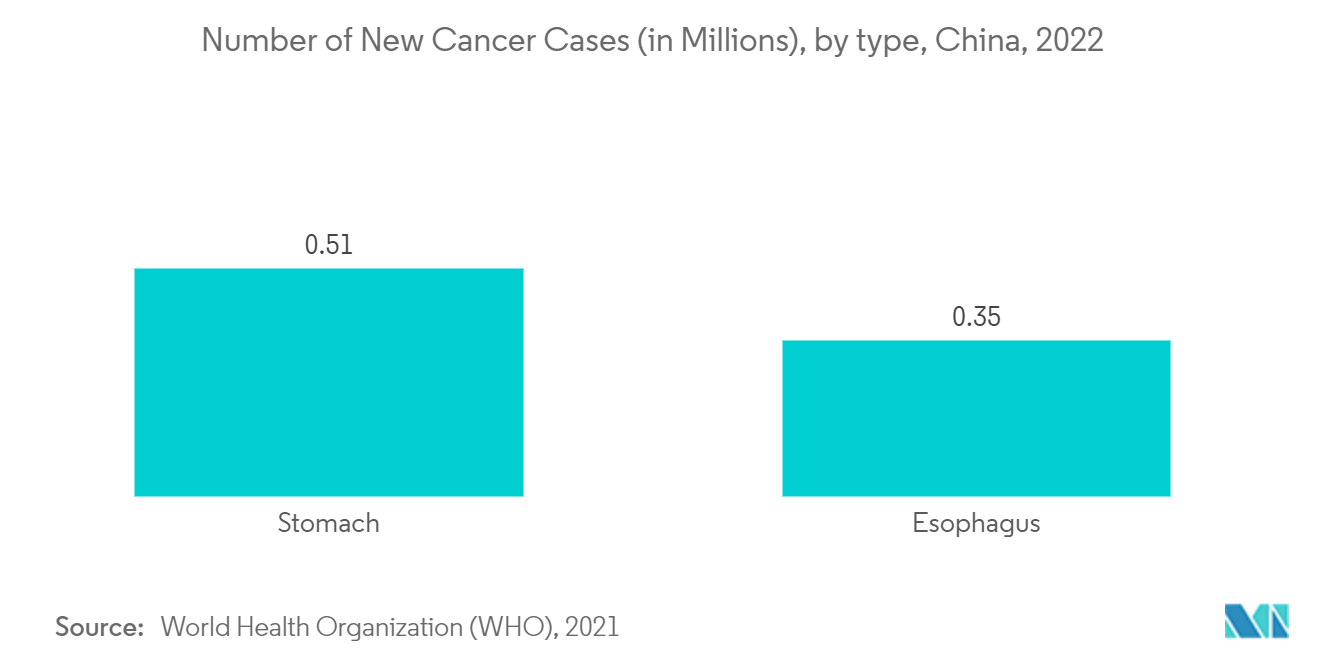
Cancer/Oncology Segment Dominates the Chinese In Vitro Diagnostics Market
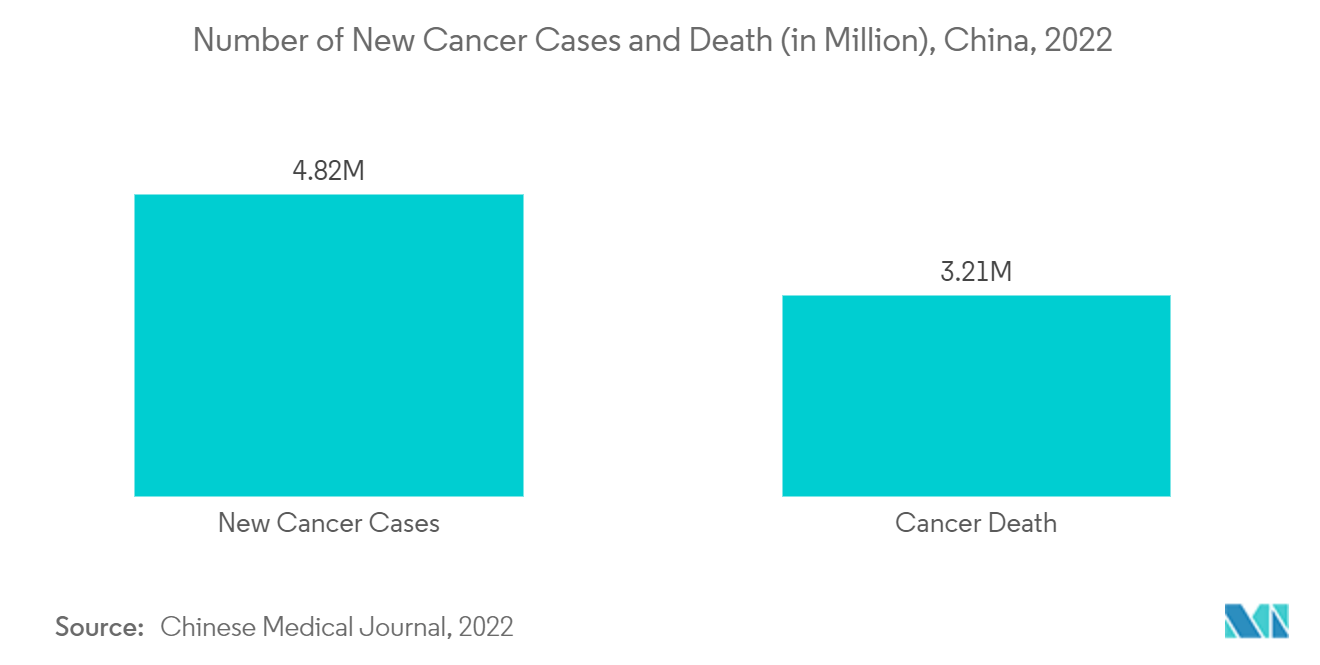
Cancer is the second-leading cause of mortality in China. Early detection of cancer and access to effective anti-cancer treatment can result in higher survival rates and a better quality of life. In vitro diagnostics (IVDs), essential in medical diagnostic testing, are tests or medical devices that examine human body specimens and provide critical data for screening, diagnosis, and treatment.
The main drivers of this market segment growth in China are the rising number of people with cancer, the increasing number of products being approved and launched, the rising amount of investments, and increasing China IVD sales.
According to the report titled "Cancer Statistics in China and the United States, 2022: Profiles, Trends, and Determinants," published in February 2022, compared to the 4,689,754 new cancer cases in China in 2020, the predicted number of new cancer cases for 2022 is 4,820,000. This increase in new cases will directly increase the demand for early diagnosis, including the use of diagnostic biomarkers, thus helping the market grow during the study period. Also, as per the report titled "Changing profiles of cancer burden worldwide and in China: a secondary analysis of the global cancer statistics 2020," published in April 2021, China is expected to experience 6.85 million new cancer cases and 5.07 million deaths in 2040. So, over the next few years, the market will be driven by the rise in cancer cases in China.
Additionally, the growing number of product launches in the region and the high concentration of key China diagnostics manufacturers act as major factors in driving the market. For instance, in August 2022, the cervical cancer screening test GynTect will be available to physicians and patients in China, the world's most populous country. In China, this is the first methylation test that has been approved for triage of HPV-positive cases, marking a significant advancement in diagnostic assays.
Because of all these factors, the market segment is expected to experience substantial growth in the China in-vitro diagnostics market over the next few years.
China In-vitro Diagnostics (IVD) Industry Overview
The China IVD market is moderately competitive and consists of some major players. Many China diagnostics suppliers in this market are trying to expand their product portfolios to increase their global market share. These companies focus on strategies such as mergers and acquisitions, collaborations, and new product developments. Key China diagnostics manufacturers in the China in vitro diagnostics market are Abbott Laboratories, Shanghai Kehua Bio-Engineering Co., Ltd., Autobio Diagnostics Co., Mindray Medical International Limited, and Maccura Biotechnology, among others.
China In-vitro Diagnostics (IVD) Market Leaders
-
Autobio Diagnostics
-
Mindray Medical International Limited
-
Shanghai Kehua Bio-Engineering Co. Ltd.
-
Maccura Biotechnology
-
Xiamen Boson Biotech
*Disclaimer: Major Players sorted in no particular order
China In-vitro Diagnostics (IVD) Market News
- September 2023: Roche Diagnostics China Technical Innovation Center (TIC) was opened in Shanghai's Pudong New Area in Jinqiao. It consists of laboratories and classrooms, covering 7,080 square meters focusing on a comprehensive upgrade of smart diagnostics, service innovation, talent training, and customer experience in diagnostics.
- 2023: Hainan government issued new guidelines regarding the administration of urgently needed imported drugs and medical devices in the Boao Lecheng International Medical Tourism Pilot Zone of the Hainan Free Trade Port.
China In-vitro Diagnostics (IVD) Market Report - Table of Contents
1. INTRODUCTION
1.1 Study Assumptions and Market Definition
1.2 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
4.1 Market Overview
4.2 Market Drivers
4.2.1 High Burden of Chronic Diseases and Infectious Diseases
4.2.2 Increasing Use of Point-of-Care (POC) Diagnostics Spurring the IVD Market
4.2.3 Increasing Awareness and Acceptance of Personalized Medicine
4.3 Market Restraints
4.3.1 Lack of Proper Reimbursement
4.3.2 Stringent Regulatory Framework
4.4 Porter's Five Forces Analysis
4.4.1 Threat of New Entrants
4.4.2 Bargaining Power of Buyers/Consumers
4.4.3 Bargaining Power of Suppliers
4.4.4 Threat of Substitute Products
4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value - USD)
5.1 By Test Type
5.1.1 Clinical Chemistry
5.1.2 Molecular Diagnostics
5.1.3 Immunodiagnostics
5.1.4 Hematology
5.1.5 Other Types
5.2 By Product
5.2.1 Instrument
5.2.2 Reagent
5.2.3 Other Products
5.3 By Usability
5.3.1 Disposable IVD Devices
5.3.2 Reusable IVD Devices
5.4 By Application
5.4.1 Infectious Disease
5.4.2 Diabetes
5.4.3 Cancer/Oncology
5.4.4 Cardiology
5.4.5 Autoimmune Disease
5.4.6 Nephrology
5.4.7 Other Applications
5.5 By End-User
5.5.1 Diagnostic Laboratories
5.5.2 Hospitals and Clinics
5.5.3 Other End-Users
6. COMPETITIVE LANDSCAPE
6.1 Company Profiles
6.1.1 Autobio Diagnostics Co.
6.1.2 F. Hoffmann-La Roche AG
6.1.3 Becton Dickinson & Company
6.1.4 Shanghai Kehua Bio-Engineering Co. Ltd
6.1.5 Abbott Laboratories
6.1.6 Thermofisher Scientific Inc.
6.1.7 QIAGEN NV
6.1.8 Xiamen Boson Biotech Co. Ltd
6.1.9 Maccura Biotechnology Co. Ltd
6.1.10 BGI Group
6.1.11 BioMerieux SA
6.1.12 Bio-Rad Laboratories Inc.
6.1.13 Danaher Corporation
6.1.14 Arkray Inc.
6.1.15 Mindray Medical International Limited
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
China In-vitro Diagnostics (IVD) Industry Segmentation
As per the scope of this report, in vitro diagnostics involves medical devices and consumables that are utilized to perform in vitro tests on various biological samples. They are used to diagnose various medical conditions, such as chronic diseases. The China in vitro diagnostics market is segmented by test type (clinical chemistry, molecular diagnostics, immunodiagnostics, hematology, and other types), product (instruments, reagents, and other products), usability (disposable IVD devices and reusable IVD devices), application (infectious disease, diabetes, cancer/oncology, cardiology, autoimmune disease, nephrology, and other applications), and end user (diagnostic laboratories, hospitals, and clinics, and other end users). The report offers the value in USD for the above segments.
| By Test Type | |
| Clinical Chemistry | |
| Molecular Diagnostics | |
| Immunodiagnostics | |
| Hematology | |
| Other Types |
| By Product | |
| Instrument | |
| Reagent | |
| Other Products |
| By Usability | |
| Disposable IVD Devices | |
| Reusable IVD Devices |
| By Application | |
| Infectious Disease | |
| Diabetes | |
| Cancer/Oncology | |
| Cardiology | |
| Autoimmune Disease | |
| Nephrology | |
| Other Applications |
| By End-User | |
| Diagnostic Laboratories | |
| Hospitals and Clinics | |
| Other End-Users |
China In-vitro Diagnostics (IVD) Market Research FAQs
How big is the China In Vitro Diagnostics Market?
The China In Vitro Diagnostics Market size is expected to reach USD 17.76 billion in 2024 and grow at a CAGR of 5.01% to reach USD 22.68 billion by 2029.
What is the current China In Vitro Diagnostics Market size?
In 2024, the China In Vitro Diagnostics Market size is expected to reach USD 17.76 billion.
Who are the key players in China In Vitro Diagnostics Market?
Autobio Diagnostics, Mindray Medical International Limited, Shanghai Kehua Bio-Engineering Co. Ltd., Maccura Biotechnology and Xiamen Boson Biotech are the major companies operating in the China In Vitro Diagnostics Market.
What years does this China In Vitro Diagnostics Market cover, and what was the market size in 2023?
In 2023, the China In Vitro Diagnostics Market size was estimated at USD 16.87 billion. The report covers the China In Vitro Diagnostics Market historical market size for years: 2021, 2022 and 2023. The report also forecasts the China In Vitro Diagnostics Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
What are the key growth areas in the China IVD Market?
The key growth areas in the China IVD Market are a) Molecular diagnostics b) Immunoassays c) POCT (point-of-care testing) for rapid diagnoses
What are the key growth drivers for the IVD market in China?
The key growth drivers for the IVD market in China are a) Rising healthcare spending b) Aging population c) Increasing focus on early disease detection
China In-vitro Diagnostics Industry Report
The China In-vitro Diagnostics industry is experiencing significant growth, driven by the increasing demand for diagnostic solutions and the rising prevalence of chronic and infectious diseases. This growth is reflected in the market size and market trends, which show a shift towards preventive healthcare across various end-users, including hospitals, homecare, research institutes, and laboratories. The industry analysis highlights the need for enhanced healthcare facilities, while industry trends indicate opportunities in rapidly growing segments.
Market research and market report data from Mordor Intelligence™ provide a comprehensive industry overview and industry report, showcasing market share and market growth. The competitive landscape reveals that players are adopting organic growth strategies to strengthen their presence, supported by government initiatives aimed at improving healthcare outcomes. The market forecast and market outlook are positive, with industry sales and industry size projected to increase.
The industry information and industry statistics underline the expansion driven by advancements in molecular diagnostics for early disease detection. Stakeholders in the healthcare sector can benefit from the market segmentation and market value insights provided in the report pdf. The report example from Mordor Intelligence™ offers detailed market review and market predictions, ensuring that research companies and other stakeholders are well-informed.
In summary, the China In-vitro Diagnostics market is poised for substantial growth, with market data and industry research indicating a favorable industry outlook. Market leaders are leveraging innovation to meet diverse end-user needs, and the market segmentation highlights the various applications driving this growth. The industry reports and market overview provide a clear understanding of the market dynamics, ensuring that stakeholders can capitalize on the opportunities presented by this evolving market.



