Cervical Total Disc Replacement (CTDR) Market Analysis
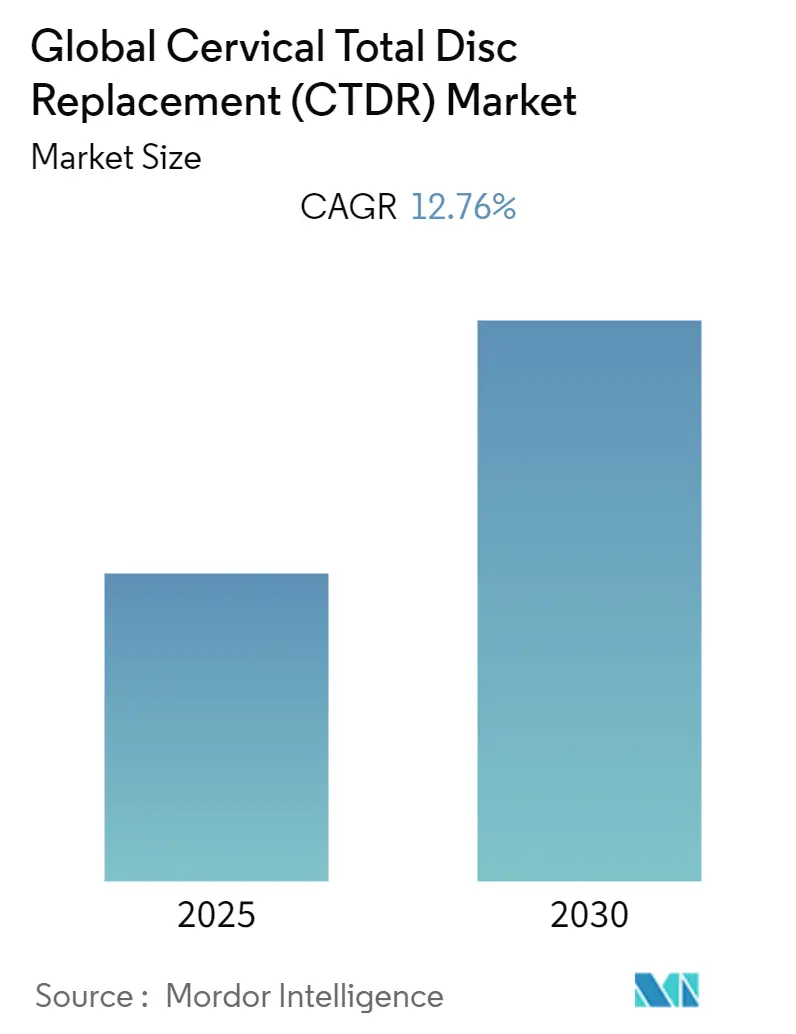
The Global Cervical Total Disc Replacement Market is expected to register a CAGR of 12.76% during the forecast period.
The COVID-19 pandemic has impacted healthcare operations, causing non-essential treatments and procedures to be suspended or delayed. In both orthopedic and spine surgery, there has been a significant decrease since the start of the COVID-19 global health crisis. In April 2020 the North American Spine Society (NASS) published a guidance sheet containing clinical recommendations and considerations for surgeries and procedures falling into the categories of urgent, urgently needed, and elective. To make the most of the available bed space, save personal protective equipment, and reduce unnecessary exposure. Elective surgeries like spinal fusion, complete joint replacements, and chronic joint issues have been postponed and delayed since the start of COVID-19. Thus, due to the above-mentioned factors, COVID-19 has significantly impacted the cervical total disc replacement device market.
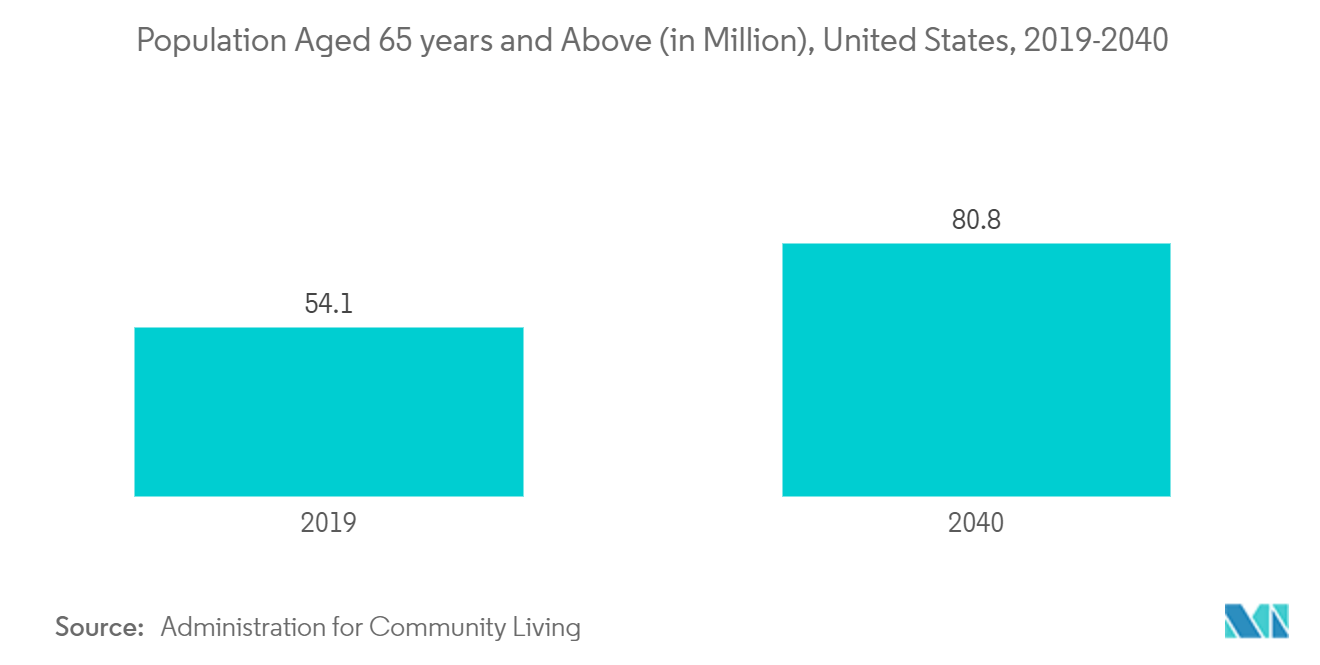
An increase in cases of cervical degenerative disc disease, the surge in the geriatric population, and the low revision surgery rate of cervical disc replacement surgery are expected to drive the cervical total disc replacement market over the forecast period. For instance, according to the Agency for Healthcare Research and Quality article published in July 2021, cervical degeneration, which increases with age, should become more common as the proportion of the population over 60 years old rises. Every year, cervical degeneration is detected in about 27% of Medicare beneficiaries. This shows the significant amount of cervical degenerative cases in hospitals resulting in a rise in cervical disc replacement surgeries. This is expected to drive the cervical disc device market. The low revision surgery rate of cervical disc replacement surgery is the other growth driving factor. In March 2021, National Library of Medicine published a systematic review article with a minimum follow-up of five years, according to the article the long-term revision rate at the index level of unsuccessful cervical total disc replacement surgery was 3.9 %.
Additionally, the advancements in technology and increasing product approvals, along with company and product acquisitions by key players are anticipated to propel the market growth. For instance, in February 2019, Orthofix Medical, a global medical device company focused on musculoskeletal products and therapies, received approval from United States Food and Drug Administration (FDA) for the M6-C artificial cervical disc, which was developed by Spinal Kinetics, a company acquired by Orthofix in April 2018. Furthermore, in February 2021 NuVasive the leader in spine technology innovation acquired Simplify Medical, the developer of the Simplify Cervical Artificial Disc (Simplify Disc) for cervical total disc replacement (cTDR). This acquisition has strengthened the cervical total disc portfolio of the company. Thus, owing to rise in product approvals and acquisitions, the studied market is expected to have significant growth over the forecast period.
However, alternative treatment options like special exercises and physiotherapy, the risk of damage to nerves or blood vessels during the surgery, and the high cost associated with cervical total disc replacement are likely to impede the market growth.
Cervical Total Disc Replacement (CTDR) Market Trends
Metal on a Biocompatible Material (M-o-B) is Expected to Witness Significant Growth Over the Forecast Period
Metal on a Biocompatible Material (M-o-B) such as titanium, tantalum, zirconium, cobalt-nickel-chromium-molybdenum, and chromium-cobalt-molybdenum are widely used in artificial disc surgeries. In terms of biocompatibility, corrosion resistance, and Young's modulus, titanium is superior. The selection of biomaterials used is based on their physiologic resistance to mechanical loading, biocompatibility, and appropriate durability. Due to its biocompatibility and super corrosion resistance properties, this type of segment accounted for the highest market growth over the forecast period.
In March 2020, the National Library of Medicine published an article stating that the ProDisc-C implants are made of an alloy of cobalt, chromium, and molybdenum with ultra-high molecular weight polyethylene and a rough titanium surface coating. In the late stages of recovery from surgery, ProDisc-C has been observed to return the complete neck's range of motion to its preoperative condition. It was also claimed that patients with ProDisc-C replacement had segmental degenerative kyphosis greatly rectified.
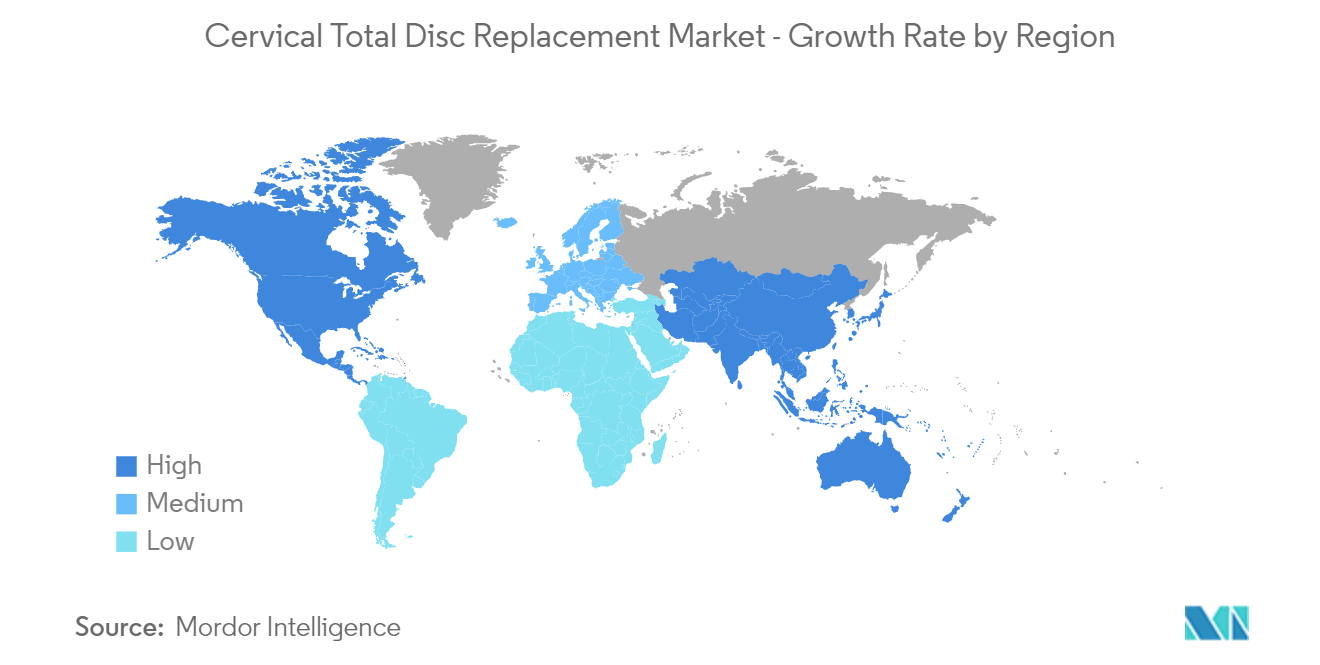
North America is Expected to Dominate the Cervical Total Disc Replacement Market
North America is expected to dominate the cervical disc replacement market owing to high in the usage of devices and the increase in the prevalence of cervical degenerative disorders in the region. For instance, according to the Spine Center at Bone & Joint Clinic of Baton Rouge, one or more spinal discs have deteriorated in about 40% of persons over the age of 40. The common degenerative cervical disorders in North America include cervical spinal stenosis, cervical disc herniation, and cervical spondylosis.
The high concentration of market players and approvals of advanced cervical disc in the United States is some of the factors driving the growth of the cervical disc replacement devices market in the country. In April 2021, NuVasive received approval from the United States Food and Drug Administration (FDA) for Simplify Cervical Artificial Disc (Simplify Disc) for two-level cervical total disc replacement (cTDR). These continuous product launches and approvals in the region are anticipated to drive the growth of the market in the region.
Therefore, owing to the aforesaid factors the growth of the studied market is anticipated in the North America Region.
Cervical Total Disc Replacement (CTDR) Industry Overview
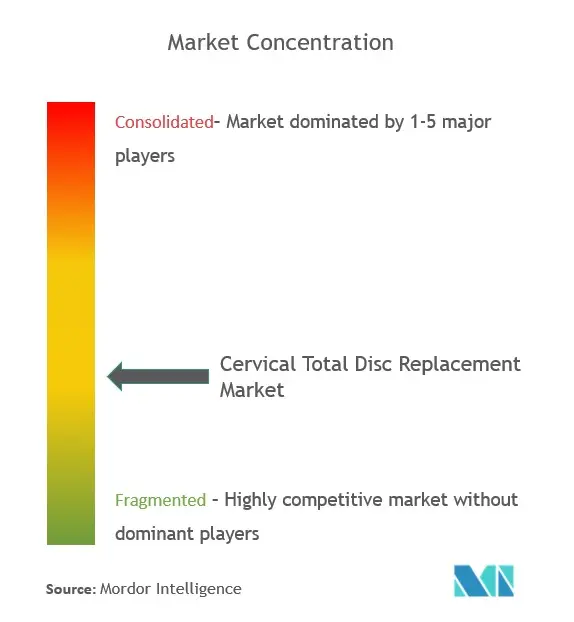
The cervical total disc replacement market is fragmented in nature due to the presence of several companies. In terms of market share, a few of the major players are currently dominating the market such as Medtronic, Plc, NuVasive, Inc., Orthofix Medical Inc., AxioMed LLC, Stryker Corporation, Zimmer Biomet Holdings, Inc, Globus Medical, Inc, Osimplant, Spine Innovations, and Centinel Spine, Inc.
Cervical Total Disc Replacement (CTDR) Market Leaders
-
Medtronic
-
AxioMed LLC
-
Globus Medical
-
NuVasive, Inc
-
Spine Innovations
- *Disclaimer: Major Players sorted in no particular order
Cervical Total Disc Replacement (CTDR) Market News
- In August 2021, Orthofix Medical announced the launch of M6-C artificial cervical disc compared to anterior cervical discectomy and fusion (ACDF) for the treatment of contiguous two-level symptomatic cervical radiculopathy in the United States.
- In September 2020, Simplify Cervical Artificial Disc (Simplify Disc) received approval from the United States Food and Drug Administration (FDA) for one-level cervical total disc replacement (cTDR).
Cervical Total Disc Replacement (CTDR) Industry Segmentation
As per the scope of the report, Cervical total disc replacement is intended to treat neck pain and neurological symptoms associated with degeneration of intervertebral discs in the cervical spine. The Cervical Total Disc Replacement Market is segmented by Material Type (Metal on a Biocompatible Material (M-o-B) and Metal on Metal (M-o-M), End User (Hospitals, Ambulatory Surgical Centers, Other End Users), and Geography (North America, Europe, Asia-Pacific, South America and Rest of the World). The report offers the value (in USD million) for the above segments.
| By Material Type | Metal on a Biocompatible Material (M-o-B) | ||
| Metal on Metal (M-o-M) | |||
| By End User | Hospitals | ||
| Ambulatory Surgical Centers | |||
| Other End Users | |||
| Geography | North America | United States | |
| Canada | |||
| Mexico | |||
| Europe | Germany | ||
| United Kingdom | |||
| France | |||
| Italy | |||
| Spain | |||
| Rest of Europe | |||
| Asia-Pacific | China | ||
| Japan | |||
| India | |||
| Australia | |||
| South Korea | |||
| Rest of Asia-Pacific | |||
| Rest of the World | |||
Cervical Total Disc Replacement (CTDR) Market Research FAQs
What is the current Global Cervical Total Disc Replacement (CTDR) Market size?
The Global Cervical Total Disc Replacement (CTDR) Market is projected to register a CAGR of 12.76% during the forecast period (2025-2030)
Who are the key players in Global Cervical Total Disc Replacement (CTDR) Market?
Medtronic, AxioMed LLC, Globus Medical, NuVasive, Inc and Spine Innovations are the major companies operating in the Global Cervical Total Disc Replacement (CTDR) Market.
Which is the fastest growing region in Global Cervical Total Disc Replacement (CTDR) Market?
Asia-Pacific is estimated to grow at the highest CAGR over the forecast period (2025-2030).
Which region has the biggest share in Global Cervical Total Disc Replacement (CTDR) Market?
In 2025, the North America accounts for the largest market share in Global Cervical Total Disc Replacement (CTDR) Market.
What years does this Global Cervical Total Disc Replacement (CTDR) Market cover?
The report covers the Global Cervical Total Disc Replacement (CTDR) Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Global Cervical Total Disc Replacement (CTDR) Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Our Best Selling Reports
Global Cervical Total Disc Replacement (CTDR) Industry Report
Statistics for the 2025 Global Cervical Total Disc Replacement (CTDR) market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Global Cervical Total Disc Replacement (CTDR) analysis includes a market forecast outlook for 2025 to 2030 and historical overview. Get a sample of this industry analysis as a free report PDF download.