Market Size of Breast Cancer Liquid Biopsy Industry
| Study Period | 2019 - 2029 |
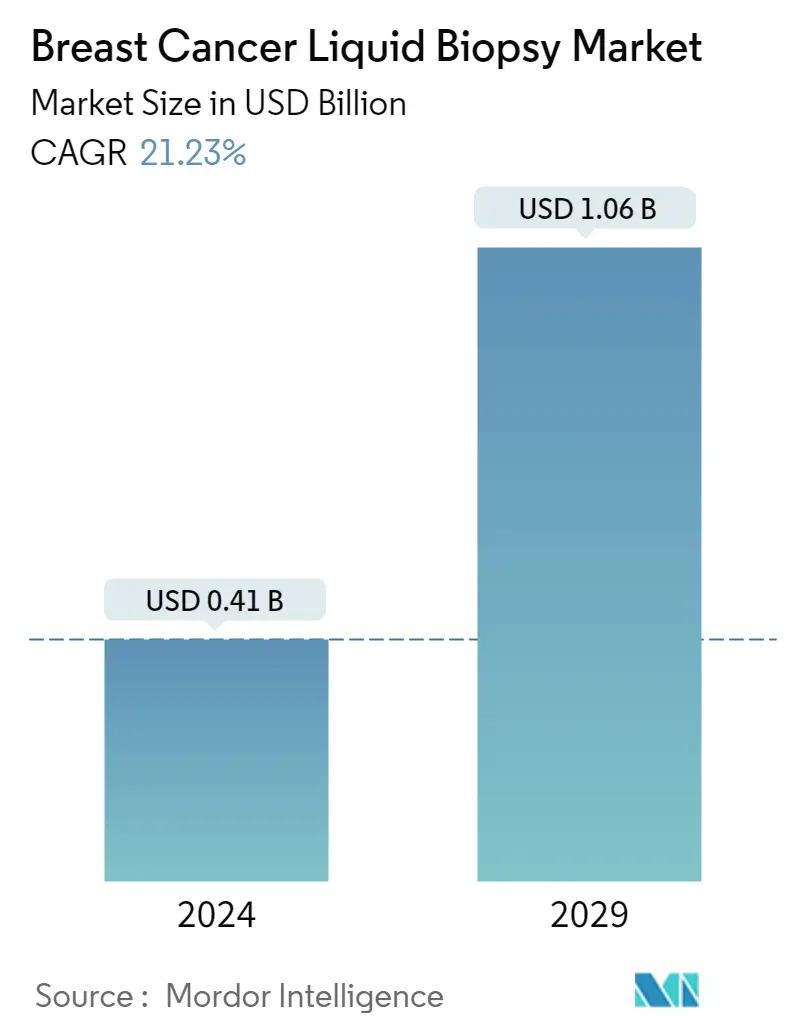
| Market Size (2024) | USD 0.41 Billion |
| Market Size (2029) | USD 1.06 Billion |
| CAGR (2024 - 2029) | 21.23 % |
| Fastest Growing Market | Asia Pacific |
| Largest Market | North America |
| Market Concentration | Medium |
Major Players*Disclaimer: Major Players sorted in no particular order |
Breast Cancer Liquid Biopsy Market Analysis
The Breast Cancer Liquid Biopsy Market size is estimated at USD 0.41 billion in 2024, and is expected to reach USD 1.06 billion by 2029, growing at a CAGR of 21.23% during the forecast period (2024-2029).
The COVID-19 pandemic is expected to have a negative effect on the market growth. The pandemic had brought several adverse impacts on breast cancer biopsy services. Hospitals, clinics, and diagnostic centers were unable to provide biopsy services due to lockdowns across some countries to prevent the spread of COVID-19. In a study conducted in the United Kingdom and published in The Lancet Oncology in March 2021, the consequences of delayed diagnosis of cancer due to COVID-19 were researched upon. Data from over 100,000 patients with breast, colorectal, esophageal, or lung cancer were evaluated in the study. The study showed that 1- and 5-year survival rates due to cancer will significantly reduce due to the pandemic because of the lack of access to screening services. Thus, as the lockdowns drag on and screening services get affected, survivability due to cancer will decrease.
An increase in regulatory approvals is expected to have a positive effect on the market. For example, in June 2019, the United States Food and Drug Administration (FDA) provided regulatory approval to QIAGEN's therascreen PIK3CA RGQ PCR Kit. The approval was granted with the device's use as a companion diagnostic assay to detect PIK3CA mutations in breast cancer for both tissue and liquid biopsies.
For the detection of breast cancer, traditional methods like tissue biopsies are not comprehensive enough to predict or capture the entire genomics landscape of breast tumors. Liquid biopsy as an alternative method for the detection of breast cancer allows many clinical applications that include screening, therapy selection, detection of mutations, disease prognosis, and drug response and resistance in patients who are unable to undergo an invasive tumor biopsy. Liquid biopsies provide unique benefits compared to other genetic testing methods, such as collecting samples with tumor biopsies. Collecting samples from the blood is a comparatively less invasive procedure and can provide a better understanding of cancer. Usually, taking a biopsy from one side of a tumor rather than the other may impact the accuracy of the results, which does not happen with liquid biopsies. Additionally, a major advantage of liquid biopsy is the possibility to detect a tumor smaller than the ones detectable by traditional screening based on imaging techniques. In fact, liquid biopsy can analyze cancer DNA, not its mass, enabling earlier tumor diagnosis.
One of the major factors leading to the market growth is the global increase in breast cancer cases. According to the World Health Organization (WHO), breast cancer is the most frequent type of cancer among women across the globe. Approximately 2.1 million women suffer from breast cancer every year, and it leads to the highest number of cancer-related deaths among women. In 2018, it was estimated that approximately 627,000 women died from breast cancer, which represented nearly 15% of all cancer deaths among women globally. According to Breast Cancer Care WA Incorporated, it was estimated that in 2019, 19,371 Australian women were diagnosed with breast cancer or approximately 53 each day, which accounts for approximately 14% of all new cancer cases diagnosed. Furthermore, according to Cancer Research UK, breast cancer is the most common cancer in the United Kingdom, accounting for 15% of all new cancer cases. The high prevalence of breast cancer and increasing demand for better minimally invasive therapies are the major driving factors in the breast cancer liquid biopsy market.
Breast Cancer Liquid Biopsy Industry Segmentation
Liquid biopsy refers to the process of obtaining tumor-derived materials such as RNA, tumor DNA, intact tumor cells, and extracellular vesicles from body fluids such as urine, blood, saliva, and stools. The procedure is relatively non-invasive, making it an attractive investigative modality as compared to traditional tumor biopsies. Breast cancer liquid biopsy is the method of detecting or investigating tumor cells in the blood sample obtained from the target cancerous site. The sample is tested for cancerous cells or biomarkers circulating in the blood.
The breast cancer liquid biopsy market is segmented by circulating biomarkers into circulating tumor cells (CTCs), circulating cell-free DNA (cfDNA), extracellular vesicles, and other circulating biomarkers. By geography, the market has been segmented into North America, Europe, Asia-Pacific, Middle-East and Africa, and South America. The report offers the value (USD million) for the above segments.
| By Circulating Biomarkers | |
| Circulating Tumor Cells (CTCs) | |
| Circulating Cell-free DNA (cfDNA) | |
| Extracellular Vesicles | |
| Other Circulating Biomarkers |
| Geography | ||||||||
| ||||||||
| ||||||||
| ||||||||
| ||||||||
|
Breast Cancer Liquid Biopsy Market Size Summary
The breast cancer liquid biopsy market is poised for significant growth, driven by the increasing prevalence of breast cancer globally and the demand for minimally invasive diagnostic methods. Liquid biopsy offers a less invasive alternative to traditional tissue biopsies, allowing for comprehensive genomic profiling and earlier tumor detection through blood sample analysis. This method is particularly beneficial for patients who cannot undergo invasive procedures, providing insights into therapy selection, mutation detection, and disease prognosis. The market is also influenced by regulatory approvals, such as the FDA's endorsement of diagnostic assays, which enhance the credibility and adoption of liquid biopsy technologies. Despite the challenges posed by the COVID-19 pandemic, which disrupted biopsy services and delayed cancer diagnoses, the market is expected to recover and expand, supported by technological advancements and increased awareness of breast cancer screening.
North America is anticipated to dominate the breast cancer liquid biopsy market, attributed to the high incidence of breast cancer and the region's robust healthcare infrastructure. The adoption of advanced diagnostic technologies like PCR and NGS is prevalent, facilitating the growth of non-invasive diagnostic methods. Key players in the market, including Qiagen, Bio-Rad Laboratories, and Biocept Inc., are actively involved in developing and offering innovative liquid biopsy solutions. These companies are enhancing their product offerings through strategic partnerships and regulatory approvals, further driving market expansion. The market's competitive landscape is characterized by a few major players holding significant market shares, while ongoing research and development efforts continue to introduce more sensitive and accurate screening techniques, promising a dynamic growth trajectory during the forecast period.
Breast Cancer Liquid Biopsy Market Size - Table of Contents
-
1. MARKET DYNAMICS
-
1.1 Market Overview
-
1.2 Market Drivers
-
1.2.1 Rising Prevalence of Breast Cancer
-
1.2.2 Increasing Demand for Better Minimally Invasive Therapies
-
1.2.3 Technological Advancements in the Early Stage Detection of Breast Cancer
-
-
1.3 Market Restraints
-
1.3.1 Stringent Regulatory Issues
-
1.3.2 Poor Reimbursement Policies
-
-
1.4 Porter's Five Forces Analysis
-
1.4.1 Threat of New Entrants
-
1.4.2 Bargaining Power of Buyers/Consumers
-
1.4.3 Bargaining Power of Suppliers
-
1.4.4 Threat of Substitute Products
-
1.4.5 Intensity of Competitive Rivalry
-
-
-
2. MARKET SEGMENTATION
-
2.1 By Circulating Biomarkers
-
2.1.1 Circulating Tumor Cells (CTCs)
-
2.1.2 Circulating Cell-free DNA (cfDNA)
-
2.1.3 Extracellular Vesicles
-
2.1.4 Other Circulating Biomarkers
-
-
2.2 Geography
-
2.2.1 North America
-
2.2.1.1 United States
-
2.2.1.2 Canada
-
2.2.1.3 Mexico
-
-
2.2.2 Europe
-
2.2.2.1 Germany
-
2.2.2.2 United Kingdom
-
2.2.2.3 France
-
2.2.2.4 Italy
-
2.2.2.5 Spain
-
2.2.2.6 Rest of Europe
-
-
2.2.3 Asia-Pacific
-
2.2.3.1 China
-
2.2.3.2 Japan
-
2.2.3.3 India
-
2.2.3.4 Australia
-
2.2.3.5 South Korea
-
2.2.3.6 Rest of Asia-Pacific
-
-
2.2.4 Middle-East and Africa
-
2.2.4.1 GCC
-
2.2.4.2 South Africa
-
2.2.4.3 Rest of Middle-East and Africa
-
-
2.2.5 South America
-
2.2.5.1 Brazil
-
2.2.5.2 Argentina
-
2.2.5.3 Rest of South America
-
-
-
Breast Cancer Liquid Biopsy Market Size FAQs
How big is the Breast Cancer Liquid Biopsy Market?
The Breast Cancer Liquid Biopsy Market size is expected to reach USD 0.41 billion in 2024 and grow at a CAGR of 21.23% to reach USD 1.06 billion by 2029.
What is the current Breast Cancer Liquid Biopsy Market size?
In 2024, the Breast Cancer Liquid Biopsy Market size is expected to reach USD 0.41 billion.

