Market Trends of Biopharmaceutical CMO And CRO Industry
The Oncology Segment is Expected to Hold a Significant Share During the Forecast Period
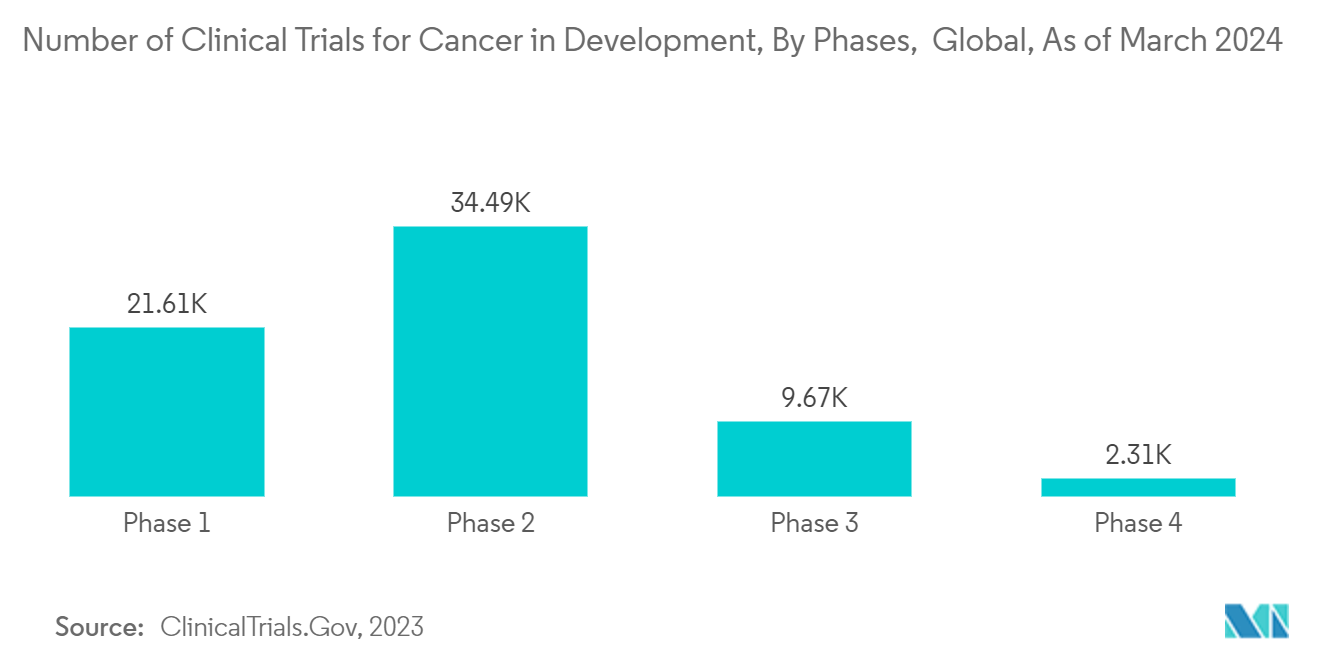
CROs (contract research organizations) play a significant role in cancer research and drug development. CROs are often involved in managing various phases of clinical trials for cancer therapies. This includes designing and planning trials, patient recruitment, data management, and monitoring trial progress to ensure adherence to regulatory standards. In addition, CRO helps pharmaceutical companies design preclinical studies, providing data analytics and interpretation and regulatory support.
The increasing prevalence of cancer is anticipated to boost the demand for advanced and effective therapeutics, leading to new investments by companies and other stakeholders, like governments, for the identification, testing, and development of novel cancer therapeutics. For instance, as per the Canadian Cancer Statistics 2023 report, about 239.2 thousand new cancer cases were reported in 2023 in Canada, compared to 233.9 thousand in 2022. Thus, the growing burden of cancer is expected to create the demand for drug research and manufacturing services, which will likely contribute to segment growth.
The increasing research & development activities and collaboration with CRO providers to effectively complete the drug research process for cancer are expected to contribute to segment growth. For instance, in June 2023, ITOCHU Corporation and its subsidiary A2 Healthcare Corporation announced a partnership agreement with NRG Oncology as a CRO to promote multicentre joint clinical research within Japan as an affiliate of the National Cancer Institutes. Based on the agreement, A2 Healthcare will continue to engage in the clinical trial support business for drugs that have yet to be approved in Japan. Additionally, in May 2023, George Clinical, a CRO, initiated preparations in Australia for two oncology Phase 1b trials that will examine a Hummingbird Bioscience precision therapy program targeting HER3, a potent driver of tumor growth and resistance against cancer drugs, in biomarker-selected patient populations, including lung cancer.
Hence, increasing research and development activities for cancer drugs will create demand for CRO services over the forecast period.
North America is Expected to Hold a Significant Market Share During the Forecast Period
North America is expected to gain the largest share in the market during the forecast period owing to the increasing incidence of chronic diseases, increasing demand for generics and biologics, rise in demand for biosimilars, and growing investments in R&D by the biopharmaceutical industry. In addition, increasing expansion of manufacturing facilities and strategic activities by the market players are expected to contribute to the market growth.
The increasing pharmaceutical expenditure in the region is also bolstering the market's growth. For instance, according to Federal Research and Development (R&D) Funding: FY2022, funding for R&D is concentrated in a few federal departments and agencies. In FY 2021, five agencies received 93.0% of total federal R&D funding, with the Department of Health and Human Services receiving 27.6%. The most significant price increases in R&D funding would be made to Health and Human Services, up to USD 7.7 billion (17.8%). These initiatives incentivize research and development activities, promote collaboration among stakeholders, and facilitate the translation of scientific discoveries into clinical applications. Contract manufacturers specializing in biopharmaceutical CMO are well-positioned to benefit from these collaborative efforts, supporting the translation of cutting-edge research into tangible healthcare solutions.
Increasing investment in developing and expanding the contract manufacturing facilities is expected to contribute to the market growth during the forecast period. CMOs have increasingly adopted continuous manufacturing for biologics production following FDA approval. For instance, in June 2022, Agilent Technologies and Abzena expanded their continuous manufacturing capabilities. Furthermore, the collaboration and partnership agreement by CDMO with the pharmaceutical company to streamline vaccine manufacturing and development are expected to contribute to market growth. For instance, in May 2023, Moderna Inc. announced a long-term agreement with Ontario-based Novocol Pharma, a CMO to perform aseptic fill-finish, labeling, and packaging of mRNA respiratory vaccines expected to be produced in Canada.
Thus, the high R&D investment and initiatives by key players in the product portfolio expansion are expected to boost the development of new drugs, increasing the demand for outsourcing services such as CMO and CRO, thereby raising the market growth.