Market Trends of Australia In-vitro Diagnostics Industry
The Molecular Diagnostics Segment is Expected to Hold a Major Market Share in the Australia In-vitro Diagnostics Market
Molecular diagnostics are a class of diagnostic tests that assess a patient's health at a cellular and molecular level. These tests detect and measure specific cellular alterations, genetic sequences in DNA or RNA, or amino acids or proteins they express. The rising number of chronic and infectious diseases in Australia is what is driving the need for molecular diagnostics.
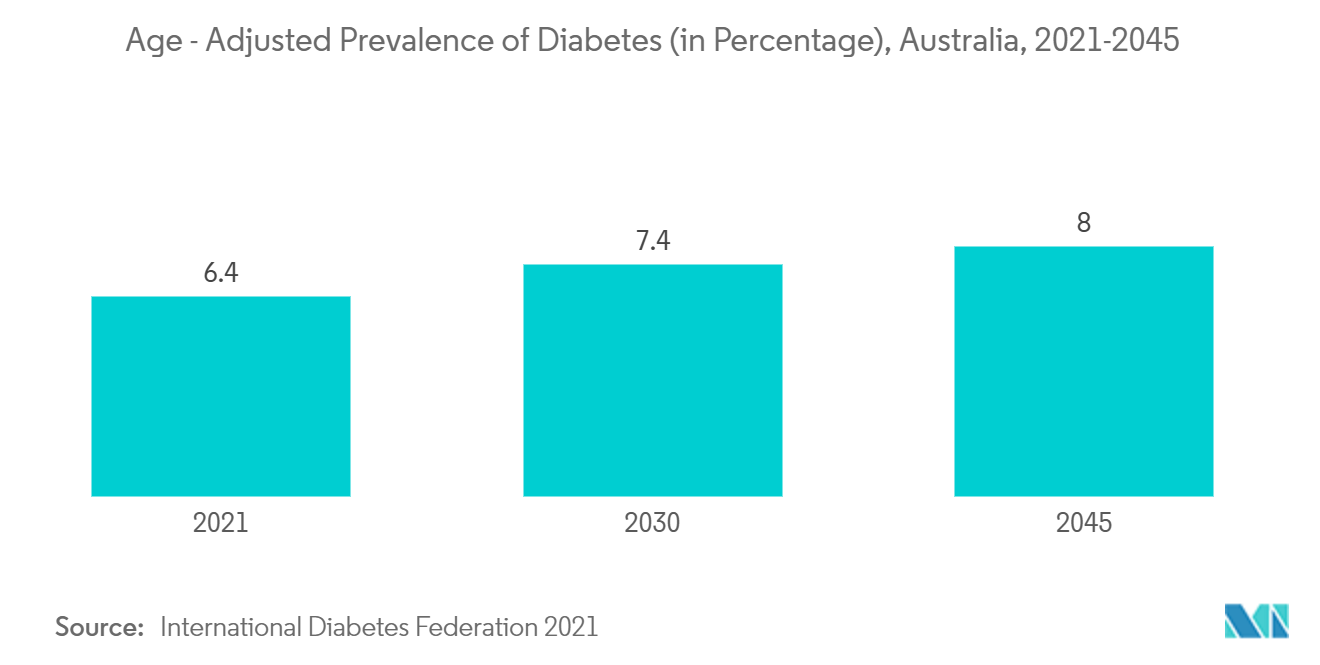
For instance, according to 2022 statistics published by the International Diabetes Federation in its 10th edition in 2021, 1.4 million people were living with diabetes in Australia. In addition, as per the same source, this number is projected to reach 1.6 million by 2030 and 1.9 million by 2050. Additionally, according to the 2022 statistics published by the Australian Institute of Health and Welfare, an estimated 571,000 Australians aged 18 and over had chronic heart diseases in 2021. Similarly, the Government of Australia data published in December 2021 mentioned that it is estimated that about 151,000 Australians were diagnosed with cancer (413 per day). The source also noted that more than 1 million people are living in Australia who are either currently living with cancer or have lived with cancer. This number is expected to grow over time as both cancer incidence and survival continue to increase. The increasing burden of various chronic diseases in Australia is expected to drive the demand for effective in-vitro diagnostics, thereby fueling the growth of the studied segment over the forecast period.
Furthermore, the increasing product approvals and research and development activities in Australia are expected to contribute to the growth of the studied segment. For instance, in April 2022, Seegene received approval from Australia's Therapeutic Goods Administration for its Allplex RV Master Assay. This assay can distinguish 21 targets for 19 different respiratory viruses, including COVID-19 and the common cold.
So, the growth of the studied segment is likely to be affected by the above-mentioned factors over the forecast period.
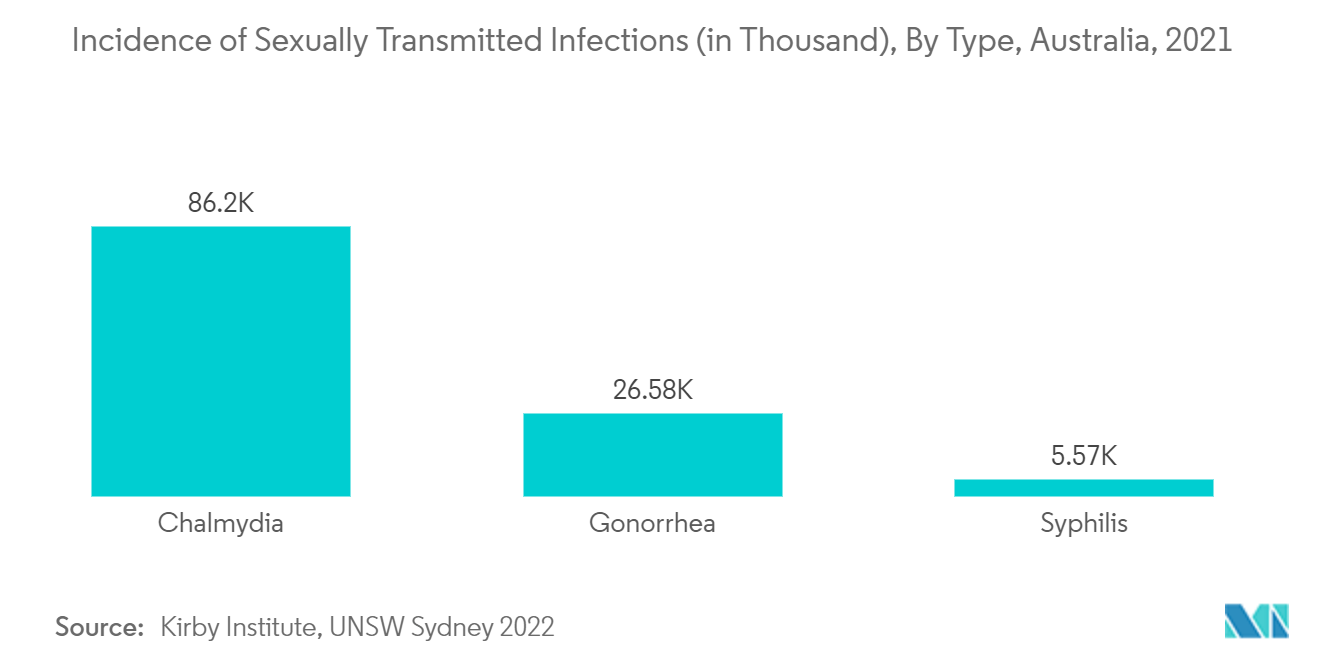
Infectious Diseases is Expected to Hold a Notable Share in the Market Over the Forecast Period
The infectious diseases segment is expected to hold a significant share of the market over the forecast period owing to the rising diseases among the Australian population, the growing demand for effective diagnostics, the presence of major market players, and frequent product launches.
For instance, the annual surveillance report published in 2022 mentioned that in 2021, chlamydia was the most frequently notified sexually transmitted infection in Australia with a total of 86916 notifications, gonorrhea cases were 26,577, and syphilis cases were 5,570. Also, the data published by the Australian Government, Department of Health and Aged Care, under the title "Sexual Health," published in 2022, mentioned that 1 in 6 Australians has had a notifiable sexually transmitted infection in their lifetime. In 2020, there were 124,900 cases of notifiable sexually transmitted infections. Such an incidence of sexually transmitted infections results in the demand for effective diagnostics, thereby contributing to the growth of the studied segment over the forecast period.
Additionally, the rising market player's strategies, such as partnerships and collaborations, along with favorable government policies, are expected to drive the segment's growth. For instance, in February 2022, Lumos Diagnostics and Planet Innovations partnered with the Melbourne state government for the shore supply of COVID-19 rapid antigen tests. The partnerships led to the creation of a USD 17.2 million rapid diagnostics manufacturing facility, equipping Victoria with the capability to make not just COVID-19 rapid tests but also tests for flu and a range of bacterial infections.
Hence, the rising infectious diseases and the growing demand for IVD, along with frequent developments in Australia for infectious diseases, are expected to boost the segment's growth over the forecast period.