Market Trends of Global Adalimumab Drugs Industry
This section covers the major market trends shaping the Adalimumab Drugs Market according to our research experts:
Rheumatoid Arthritis Dominates the Market and is Expected to Continue to Do the Same during the Forecast Period
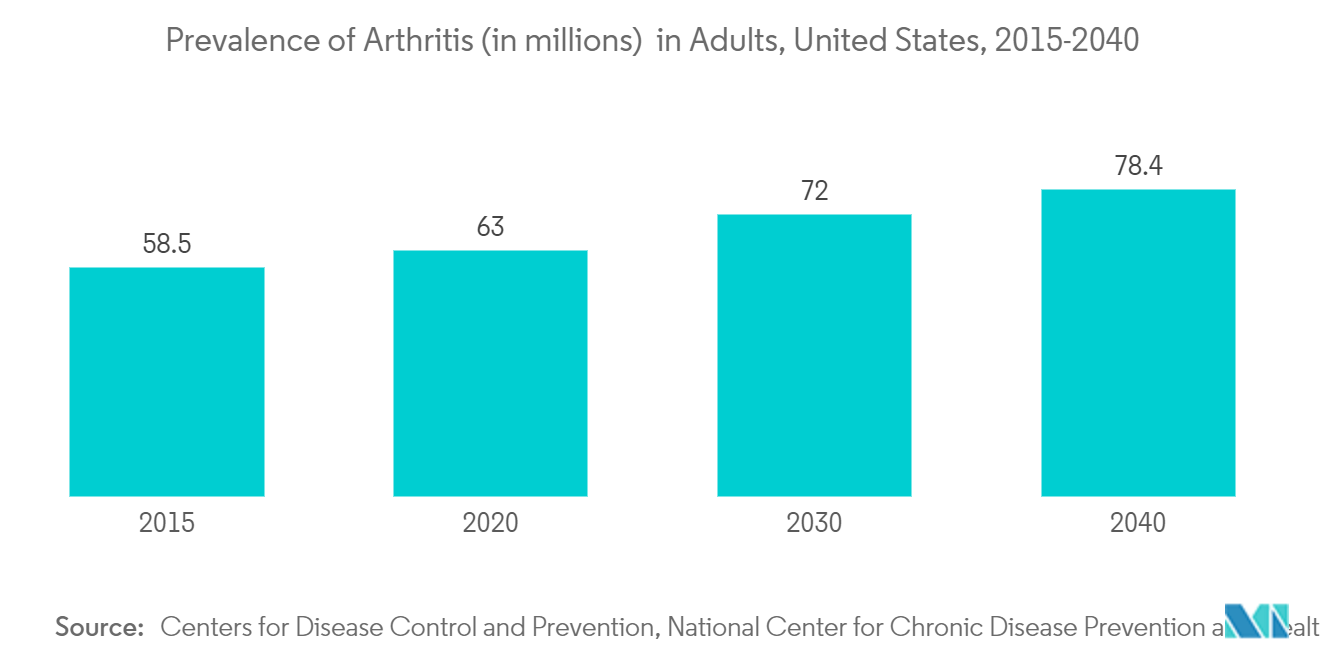
Rheumatoid arthritis is expected to dominate the global adalimumab drugs market through the forecast period. This can be attributed to the rising prevalence of rheumatoid arthritis which causes different symptoms such as joint pain, swelling, and stiffness, which are generally accompanied by chronic pain and the inability to perform daily activities. Over a prolonged period, this disorder can hamper a patient's mobility and can lead to permanent joint damage. If left untreated, this disorder can lead to mobility impairment and risk of joint replacement.
According to an article titled 'The Prevalence and Statistics of Arthritis' published in February 2022, the incidence of rheumatoid arthritis is 2 to 3 times higher in women than men. In both men and women, the onset of rheumatoid arthritis is highest among people in their 60s. Another article titled 'Epidemiology of risk factors for, and possible causes of rheumatoid arthritis' published in 2022, states that rheumatoid arthritis (RA) is estimated to affect approximately 0.24% to 1% of the population and to be twice as common in women compared with men. A range of poor long-term outcomes can potentially occur, but these have been reduced to some degree with the use of more aggressive treatment strategies and more effective drug therapies. Thus, the rheumatoid arthritis segment is witnessing a shift toward combination therapies that provide enhanced results to patients. This is expected to drive segmental growth and accelerated market growth.
As per an article titled 'How to Afford Biologics for RA' published in December 2021, types of biologics your doctor may recommend for rheumatoid arthritis includes tumor necrosis factor (TNF) inhibitors like adalimumab (Humira), certolizumab (Cimzia), etanercept (Enbrel), golimumab (Simponi), and infliximab (Remicade). Thus, the cost of the drugs to be consumed can be made of low cost by the private health insurance plans covering biologics for rheumatoid arthritis. This is expected to accelerate segmental growth.
Higher availability, positive results, and low price of a biosimilar for the target disease are supplementing the growth of the segment. The introduction of new biosimilars such as Hadlima and Adaly in the treatment of rheumatoid arthritis is poised to create a significant shift in prescription patterns.
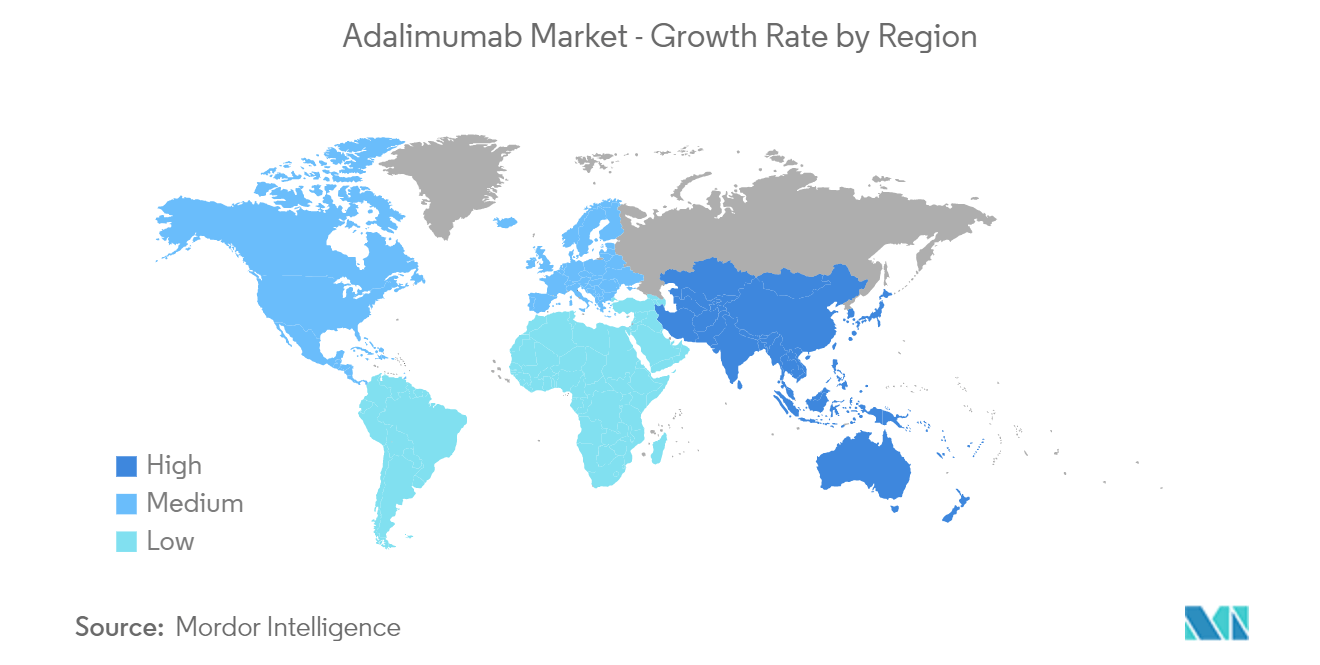
North America Represents the Largest Market and Asia-Pacific is Expected to Register Fastest Growth.
North America is at the forefront of growth in the region and is also an important revenue contributor in the global arena. Heightened awareness of disease remittance therapies among patients, rising prevalence of RA, and high public and private healthcare spending are stimulating the growth of the region. As per the World Health Organization, an estimated 54.4 million adults in the United States suffer from rheumatoid arthritis. Moreover, easy access to quality healthcare, favorable reimbursement policies, a strong clinical pipeline, and approval of novel drugs are projected to promote revenue growth in North America. For instance, in November 2021, Amgen said that its seeking interchangeability status for its low-concentration adalimumab biosimilar (Amjevita). Amjevita was approved by the FDA in September 2016 and is expected to be the first adalimumab biosimilar to launch in the United States in January 2023. In January 2022, Samsung Bioepis and Organon announced that they are seeking interchangeability for a high-concentration, citrate-free version of their adalimumab biosimilar (Hadlima), which was approved as a low-concentration formulation in July 2019 and is expected to launch on the US market in June 2023. Additionally, in October 2021, The United States Food and Drug Administration approved the first interchangeable biosimilar product to treat certain inflammatory diseases. Cyltezo (adalimumab-adbm), originally approved in August 2017, is both biosimilar to, and interchangeable with, its reference product Humira (adalimumab) for Cyltezo's approved uses.
Asia Pacific is likely to post the fastest market throughout the forecast horizon. Growing adoption of the urban lifestyle is leading to expanding base of patients in the region. This, coupled with increasing healthcare spending, is expected to propel the market in the APAC region. Favorable regulatory policies for biosimilars are estimated to boost demand for adalimumab over the forecast period.
Price-sensitive regions such as Latin America and MEA have higher acceptance of generic drugs as compared to innovator biologics due to lower prices. Increasing demand for biosimilars for disease reversal is poised to augment the regional markets during the forecast period.